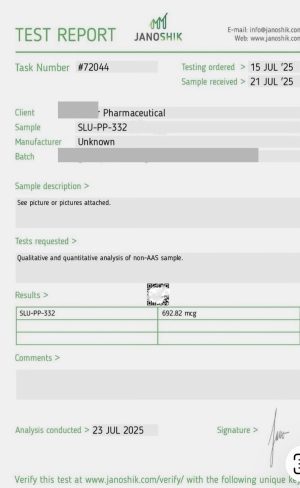
5 Amino 1 MQ 10 MG
5-Amino-1MQ (10mg) is a research compound designed to act as a potent inhibitor of the enzyme nicotinamide N-methyltransferase (NNMT). It is primarily studied for accelerating fat loss, improving metabolism, and supporting cellular energy (NAD+ levels). Standard research dosages are typically 10mg daily.
5-Amino-1MQ
Reddit user comments:
- “Unlike pure appetite suppressants, 5-Amino-1MQ works on your metabolism, helping you burn fat while preserving energy.”
- "There are times when weight loss becomes virtually impossible… one way to break this cycle is with a peptide like 5-amino-1MQ.
- My energy levels are amazing and I feel great on it.”
Some users say they notice:
easier workouts
improved stamina
less fatigue during dieting
This is often attributed in discussions to the compound’s influence on cellular metabolism.
Metabolic Research Compound Overview
5-Amino-1MQ is a research compound scientists study for its role in how the body manages energy, metabolism, and fat storage.
Researchers are interested in 5-Amino-1MQ because it interacts with a specific metabolic enzyme that helps control how the body processes fuel and stores energy.
Understanding these systems helps scientists learn more about how metabolism works.
Understanding Metabolism in Simple Terms
Your body runs on thousands of tiny chemical reactions that convert food into energy.
These reactions happen through metabolic pathways, which are basically step-by-step processes your cells use to turn nutrients into energy or store them as fuel.
You can think of metabolic pathways like an assembly line in a factory.
Each step changes a molecule slightly until the body produces something useful, such as:
energy for your cells
stored fat
hormones and signaling molecules
What Are Metabolic Enzymes?
Inside these pathways are metabolic enzymes, which are special proteins that act like workers on the assembly line.
Each enzyme performs one specific job.
For example:
1️⃣ One enzyme changes a molecule into the next step
2️⃣ Another enzyme changes that molecule again
3️⃣ Eventually the body produces energy or stores fuel
Without enzymes, these reactions would happen too slowly for the body to function.
The Enzyme Researchers Study With 5-Amino-1MQ
The enzyme most often studied with 5-Amino-1MQ is called NNMT.
This enzyme plays a role in how the body manages:
- energy balance
- fat storage
- cellular fuel usage
- metabolic efficiency
Researchers are studying how compounds that interact with NNMT may influence these metabolic systems.
Why Researchers Study 5-Amino-1MQ
Scientists exploring metabolic health are interested in compounds that help them understand how the body:
- processes nutrients
- stores energy
- regulates fat metabolism
- controls cellular fuel systems
By studying molecules like 5-Amino-1MQ, researchers can learn more about how the body’s metabolic “assembly lines” operate.
This helps expand scientific understanding of metabolism and cellular energy systems.
Related Areas of Metabolic Research
Researchers investigating metabolic pathways often study several compounds that influence energy regulation, including:
- 5-Amino-1MQ
- MOTS-C
- NAD+ related compounds
- GLP-1 pathway peptides such as semaglutide and tirzepatide
These compounds help scientists better understand how metabolic systems work inside the body.
Frequently Asked Questions
What is 5-Amino-1MQ?
5-Amino-1MQ is a research compound studied for its interaction with metabolic enzymes involved in cellular energy systems.
What is the NNMT enzyme?
NNMT is a metabolic enzyme involved in pathways related to energy balance and fuel usage in cells.
Why are researchers studying metabolic enzymes?
Metabolic enzymes control how the body processes energy and nutrients, making them important targets for scientific research.
Research Use Notice
All compounds offered by GreyVita are intended strictly for laboratory research purposes only.
These materials are not intended for human or veterinary use.

Acetic Acid 3 ML
Acetic Acid 0.3% – 10ml (Sterile Solution) – For Laboratory Use Only
Acetic Acid 0.6% is a sterile, aqueous solution commonly used in research and lab settings for pH adjustment, buffer preparation, and preservative applications. Its mild acidic properties make it suitable for studies involving topical antimicrobial effects, cellular environment modification, and reagent preparation.
This solution is intended for controlled research environments requiring low-concentration acetic acid in a sterile, injectable-grade format.
Product Details:
Compound: Acetic Acid 0.6% in sterile water
Volume: 3ml multi-dose vial
Form: Sterile solution
Concentration: 0.6% w/v
Storage: Store at 68–77°F (20–25°C); protect from light and contamination
Grade: For laboratory research only. Not for human or animal use in therapeutic or diagnostic procedures.
Potential Research Applications:
pH regulation and buffer system studies
Topical antimicrobial and cytotoxicity research
Reagent dilution and formulation preparation
Laboratory protocol requiring acidic conditions
For laboratory use only. This product is not intended to diagnose, treat, cure, or prevent any disease.

AOD9604 5MG
Research: metabolism, target fat cells
Please be aware all AOD will naturally gel over time, even when mixed with acetic acid. This is a normal characteristic of the product and does not affect its usability. In fact, gelled AOD remains fully functional and can still be used for any research application without issue.
We highly recommend using 1ML acetic acid and 2ML bacteriostatic water. If the product begins to gel simply add more acetic acid. Always reconstitute aod slowly. Using too much agitation can cause AOD to turn solid. To mitigate this issue altogether simply preload all your syringes immediately after reconstitution.
The best way to reconstitute AOD is by using 2ML of Bacteriostatic Water and then slowly add Acetic Acid. Do not splash acetic acid in but rather add a little at a time.
Our AOD is of ultra-high purity, which means it may gel faster than other formulations on the market. This is not a defect but rather a result of its exceptional quality and concentration.
What Reddit Users Say About AOD-9604
Reddit User Experiences With AOD-9604
Many Reddit users report targeted fat loss or body recomposition, particularly in the abdomen.
“AOD is working for me… I believe the AOD is reducing my belly fat. I see a lot of changes especially below the umbilicus.”
r/Peptides_for_Women
Another user said it helped maintain weight loss after stopping it:
“AOD is great if you ask me. I'm taking my first break from it right now and have maintained my loss.”
r/PeptidesForum
Others describe localized fat changes rather than overall weight loss:
“AOD has substantially targeted my stomach… it’s really decreased and thinned out.”
r/Retatrutide
Common positive themes Reddit users mention:
- Belly fat reduction
- Body recomposition rather than scale weight loss
- Works better with fasting + cardio
- Useful stack with GLP-1s
Neutral / Mixed Experiences
A lot of Reddit feedback says results are subtle or slow.
Example:
“I've read mostly positive reviews but a few negatives of it doing nothing.”
r/Tirzeglutide
Some users say it's not strong enough alone:
“It does help with reducing belly fat. I doubt it's going to do much in the weight loss without combining it with a GLP-1.”
r/PeptidesNootropics
Common neutral themes:
- Slow results (weeks or months)
- Better as a support peptide
Often stacked with: - retatrutide
- tirzepatide
- semaglutide
- LipoC with L-carnitine
- fat-burning workouts
AOD-9604 – Research Overview
Chemical name: Fragment 177–191 of human growth hormone
Common research name: AOD-9604
Class: Modified GH-fragment peptide
Sequence: Tyr-Leu-Arg-Ile-Val-Gln-Cys-Arg-Ser-Val-Lys
Molecular weight: ~1.8 kDa
Target: GH-fragment–related metabolic pathways (fat-metabolism pathways)
Molecular & Pharmacological Profile
AOD-9604 is a modified fragment of human growth hormone that has been investigated in preclinical studies (animal and cell-based research) for its effects on lipid metabolism (how fat is stored or broken down).
Unlike full-length GH, AOD-9604 does not activate growth hormone receptors (does not behave like GH) and does not increase IGF-1 levels (does not trigger growth-related hormones).
Research in animal and in-vitro models suggests that this fragment may influence:
- Lipolysis (breakdown of stored fat)
- Lipogenesis (creation of new fat)
- Fat oxidation (burning fat for energy)
All findings refer strictly to laboratory settings, not established effects in humans.
Mechanism of Action (Preclinical)
In experimental systems, AOD-9604 has been shown to:
- Increase the breakdown of stored triglycerides in adipocyte cultures (fat cells releasing stored fat)
- Reduce the formation of new lipids in adipose tissue (less new fat being created)
- Enhance lipolytic enzyme activity (enzymes that help break down fat become more active)
- Act independently of GH receptor signaling (does not activate growth pathways like regular GH)
- This activity appears to be specific to metabolic pathways in fat tissue (primarily affecting fat-related processes rather than other systems).
Preclinical Research: Potential Applications
AOD-9604 has been explored in animal and cell-based research models for:
- Metabolic research (studying how fat is stored, mobilized, or burned)
- Obesity research (understanding fat-mass changes in diet-induced obese rodents)
- Adipocyte biology (how fat cells develop, store energy, or break down lipids)
- Energy-utilization studies (how cells use fuel or shift toward fat oxidation)
These investigations relate only to preclinical experimentation, not clinical or therapeutic use.
Efficacy Observed in Preclinical Models
Rodent studies:
- AOD-9604 was associated with increased fat oxidation (burning more fat for energy)
- Reduced body fat accumulation (less fat gain) in diet-induced obese rats
Observed metabolic changes occurred without increases in IGF-1 (no growth-hormone–like effects)
In vitro (cell-culture) studies: - Fat cells showed enhanced lipolytic activity (greater breakdown of stored fat)
- Reduced lipid droplet formation (less buildup of new fat inside cells)
- Changes in gene expression related to fat storage vs. fat breakdown (shift toward burning rather than storing)
- These findings are limited to laboratory research, not human outcomes.
Safety Tolerability (Preclinical)
In animal research:
- AOD-9604 did not stimulate IGF-1 (no GH-like growth signaling)
- No increases in linear growth in young rodents (no growth-promoting activity)
- Dose-related metabolic effects were observed (changes in fat metabolism)
Long-term toxicity, human pharmacokinetics, and human tolerability remain undetermined (not studied extensively)
AOD-9604 has not been evaluated or approved as safe for human use.
Regulatory Status Research Disclaimer
- AOD-9604 is an investigational research compound.
- It is not approved by the FDA or any regulatory authority for medical, therapeutic, dietary, or cosmetic use.
- For Research Use Only.
- Not for human consumption. Not for veterinary use.
- Not for diagnostic or therapeutic purposes.
- No medical claims are made or implied.
- Researchers are responsible for compliance with all applicable regulations.
Key References (Preclinical)
Heffernan M. et al. The lipolytic effect of human growth hormone fragment 177–191 in obese Zucker rats. J Endocrinol.
Ng FM. HGH fragment 177–191 increases lipolysis in isolated rat adipocytes. Biochem Mol Biol Int.
Chen C. et al. GH-fragment-specific metabolic activity independent of IGF-1. Peptides.
Heffernan et al. Selective fat-tissue effects of GH-derived fragments. Endocrinology Research.

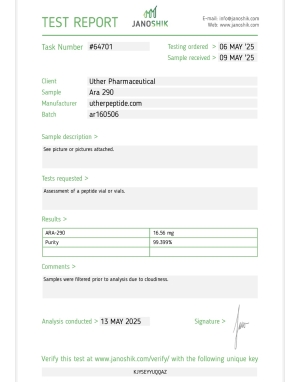
ARA-290 16MG
May require AA Water for reconstitution. Some customers report gelled consistency or cloudiness with BAC water recon.
ARA-290 (16mg) is a synthetic 11-amino acid peptide, also known as Cibinetide, derived from the tissue-protective domain of erythropoietin (EPO). It is designed for research purposes to target the Innate Repair Receptor (IRR) for treating neuropathic pain, inflammatory conditions, and promoting tissue repair, specifically targeting small-fiber neuropathy, without stimulating red blood cell production.
Reddit User Experiences With ARA-290
Many Reddit users report improvements in neuropathy symptoms, particularly sensation returning or pain decreasing.
Example from a small-fiber neuropathy discussion:
> “Feeling started to come back… I was surprised how aware I was of temperature.”
> r/smallfiberneuropathy
Another user described improved nerve sensation:
> “I could feel the fabric of my pants sliding across my skin… temperature too.”
> r/smallfiberneuropathy
Some users report meaningful reductions in nerve pain:
> “For me, it brought about a 25–50% reduction in pain.”
> r/smallfiberneuropathy
Others say it helped overall inflammation and recovery when used in a peptide stack:
> “I feel less inflamed, my digestion and sleep are better.”
> r/biohackers
Common positive themes mentioned on Reddit:
• reduced neuropathic pain
• improved sensation in feet/legs
• reduced inflammation
• better recovery after intense exercise
Key Aspects of ARA-290 16mg:
Mechanism of Action: Selectively binds to the EPOR–CD131 receptor complex (IRR), reducing pro-inflammatory cytokines, oxidative stress, and promoting tissue regeneration.
Targeted Conditions: Research focuses on its potential to improve symptoms in type 2 diabetes, sarcoidosis, and other chronic neuropathic pain conditions.
Non-Erythropoietic: Unlike EPO, it does not increase red blood cell production, avoiding associated cardiovascular risks.
Formulation: lyophilized (freeze-dried) powder requiring reconstitution for subcutaneous research application.
Research Status: It is primarily studied in preclinical models and Phase II clinical trials, with potential applications in nerve fiber regeneration (e.g., in the cornea).
Disclaimer: ARA-290 is generally sold for research purposes only (RUO) and not for human consumption, unless it is part of an approved clinical trial or specialized, compounding pharmacy prescription.


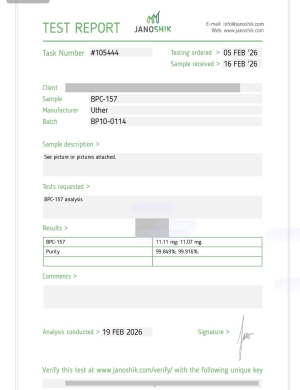
BPC-157 10MG
BPC-157 — Research Summary & Potential Benefits
BPC-157 (Body Protection Compound-157) is a synthetic peptide derived from a naturally occurring protein found in the stomach. It has been widely studied in laboratory and preclinical models for its potential roles in tissue repair, inflammation modulation, and gastrointestinal support. Research continues to explore how BPC-157 influences cellular pathways related to healing, protection, and recovery.
Reddit User Experiences With BPC-157
Many Reddit users report faster recovery from injuries, especially tendon, ligament, and muscle problems.
Example from a climbing injury discussion:
> “BPC-157 seemed to give a noticeable boost… daily pain improved.”
> r/climbharder
Another user recovering from a knee injury wrote:
> “My swelling dropped within days, and range of motion improved.”
> r/acl
Some athletes say it noticeably improves training recovery:
> “My faster recovery from workouts is really baffling.”
> r/crossfit
Other Reddit users describe general improvements in inflammation and healing:
> “Less inflammation… maybe a 20% boost in recovery.”
Common positive themes on Reddit
• tendon and ligament healing
• reduced inflammation
• faster recovery after workouts
• improved range of motion after injury
This matches the way athletes commonly use it — for soft-tissue injuries and rehabilitation.
✨ Key Research Areas
Tissue Healing & Regeneration
In preclinical studies, BPC-157 has shown the ability to accelerate repair in muscle, tendon, ligament, nerve, and skin models. Research suggests it may support angiogenesis (new blood vessel formation) and fibroblast activity, both essential for healing.
Gastrointestinal Support
Because BPC-157 originates from a gastric protein, many studies highlight its potential to support the gut lining, protect against inflammation, and aid recovery in models of gastric injury and IBS-type irritation.
Anti-Inflammatory Properties
Research shows that BPC-157 may help modulate inflammatory cytokines, reduce oxidative stress, and promote protective pathways—supporting recovery in soft tissues, joints, and vascular systems.
Neuroprotective Effects
Animal studies indicate that BPC-157 may help protect neurons, reduce the impact of injury, and influence serotonin and dopamine pathways, contributing to healthier neurological responses.
Cardiovascular & Circulatory Support
BPC-157 has been shown to promote healthy blood vessel repair and protect endothelial tissue, supporting overall vascular function in research models.
Bone & Joint Repair
Some studies demonstrate improved bone healing and protection of joint structures, potentially due to BPC-157’s effects on growth factors and pro-healing signaling pathways.

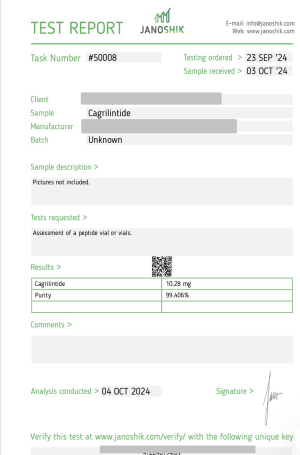
Cagri 10MG
CAS: 1415456-99-3
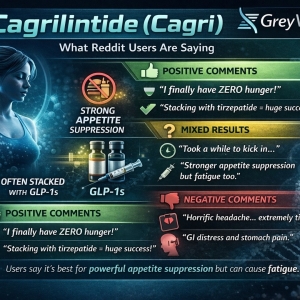
Reddit User Experiences With Cagrilintide (Cagri)
Positive Experiences
Many Reddit users report strong appetite suppression, especially when stacked with GLP-1 peptides like tirzepatide or semaglutide.
Example from a user discussing the combo with tirzepatide:
“Cagri stops any hunger for me but makes me tired.”
r/Cagrilintide
Another user who used it long-term with tirzepatide said:
“Been on the same dose of cagri/tirz for well over a year… results are worth it.”
r/Cagrilintide
People in clinical trials or research communities also report very fast appetite reduction:
“ZERO hunger… about 4 hours after my first dose.”
r/Zepbound
Common positive themes reported on Reddit:
• strong appetite suppression
• easier calorie restriction
• enhanced weight loss when stacked with GLP-1s
• reduced “food noise”
Some users even say it helps them stay on lower GLP-1 doses because the appetite effect is strong.
Cagrilintide (Cagri) – Research Overview
Chemical name: Acylated amylin analog
Class: Long-acting amylin receptor agonist
Target: AMY1/AMY2/AMY3 receptors (amylin receptor complexes)
Half-life: Extended, once-weekly profile in research models
Cagrilintide is a synthetic, stabilized analog of amylin — a naturally occurring peptide co-secreted with insulin.
It has been investigated in preclinical research for its effects on:
Appetite regulation
Gastric emptying
Body-weight modulation
Energy balance
Satiety signaling in the brain
All findings are from animal and in-vitro (lab) research.*
How Cagrilintide Works (Preclinical)
Cagri activates amylin receptors (AMY1, AMY2, AMY3), which are heavily involved in metabolic regulation.
Key mechanisms observed in research models:
- Appetite suppression via amylin-pathway signaling
(reduced desire to eat / earlier sense of fullness) - Slowed gastric emptying
(food moves more slowly through the stomach, increasing satiety) - Reduced meal size and frequency in animal models
(animals ate smaller meals and less often) - Modulation of hypothalamic satiety centers
(brain regions that regulate hunger become more responsive) - Lower food-seeking behavior in preclinical models
(reduced "food noise" in rodent behavioral studies)
Cagrilintide works through the amylin pathway, which is different from GLP-1 signaling.
This is why it behaves differently than semaglutide or tirzepatide.
Preclinical Research: Benefits of Cagrilintide By Itself
(Meaning not combined with semaglutide)
Below is a breakdown of what research has shown cagrilintide does on its own in animals and experimental systems.
- Appetite Reduction (decreased food intake)
In rodent models, Cagri produced a dose-dependent reduction in food intake
(animals ate significantly less).
This effect occurred through:
Amylin-receptor activation (satiety hormones)
Slowed gastric emptying (longer fullness)
Reduced reward-driven eating (less “hedonic feeding”) - Significant Weight-Modulating Effects in Animals
In diet-induced obese rodents:
Weight decreased more than with amylin alone
Fat mass specifically was reduced
Lean mass was more preserved (compared to some GLP-1 studies)
Cagrilintide alone produced meaningful weight-modifying effects in mice and rats without the need for GLP-1 agonists. - Very Strong Satiety Signaling
(researchers note this is stronger than semaglutide alone in certain settings)
Cagri produces:- Earlier meal termination (animals stop eating sooner)
- Greater fullness between meals
- Less interest in high-reward foods
- This is one reason why Cagri + sema is potent — but Cagri on its own is already highly effective.
- Slowed Gastric Emptying
(food digests more slowly, prolonging satiety)
Cagri has a pronounced effect on gastric motility:- Delayed stomach emptying
- Longer feelings of fullness
- Less frequent feeding behavior
This effect is mediated through amylin receptors rather than GLP-1 receptors.
- Improvements in Metabolic Markers (Preclinical)
- Cagri has shown potential preclinical effects on:
- Post-meal glucose excursions (smaller blood sugar spikes)
- Energy expenditure patterns (slight increases in metabolic rate in some models)
- Lipid metabolism (changes in fat-partitioning pathways)
Again:
These findings are animal-specific and don’t translate to confirmed human outcomes.
- Strong Effect on “Food Noise” in Rodents
(reduced obsessive or compulsive feeding behaviors)
Cagri is well-documented to impact:- Reward-driven eating
- Hyper-phagia (over-eating)
- Craving-like feeding behaviors
- In some comparison models, Cagri reduced compulsive feeding even more strongly than GLP-1 agonists.
- Long Half-Life with Once-Weekly Activity in Animals
Cagrilintide is acylated (fatty-acid–attached), allowing it to bind albumin —
(this keeps it in circulation longer).
In rodents and primates:- Single weekly dosing achieved stable levels
- Effects lasted throughout the dosing interval
- Concentration peaks were smoother (less nausea-associated spiking)
How Cagri Differs from Semaglutide (When Used Alone)
Here’s a simple breakdown:
Cagri (amylin pathway):
- STRONG satiety
- STRONG food-noise reduction
- STRONG appetite suppression
- Moderate gastric slowing
- Moderate metabolic effects
Semaglutide (GLP-1 pathway):
- Moderate-to-strong appetite suppression
- Strong gastric slowing
- Strong insulin/glucose effects
- Nausea risk tied to peaks
Cagri alone = “behavioral hunger control”
(in rodent models, animals showed less emotional/reward eating)
Sema alone = “physiological hunger control + glycemic effects”
This is why together they’re synergistic…
but Cagri alone is still a powerful satiety- and appetite-modulating peptide in its own right.
Safety & Tolerability (Preclinical)
- Nausea-like behavior in rodents was lower than in some GLP-1 models
- No acute toxicity was seen at research doses
- Amylin-pathway effects were dose-dependent
- Long-term safety, human pharmacokinetics, and clinical tolerability remain undetermined
Regulatory Status
Cagrilintide is an investigational research compound.
Not approved for medical, therapeutic, dietary, or cosmetic use.
For Research Use Only.
Not for human consumption.
Not for veterinary use.
No medical claims are made or implied.
It has been studied for benefits not just in type 2 diabetes, but for liver damage, alcohol-related liver disease, and heart/blood vessel disease.
There is some speculation about the role of this peptide in Alzheimer’s disease as well, but no research has been published in that particular sub-domain, yet.
Many trials, however, have looked at the combination of cagrilintide and semaglutide in the treatment of obesity and type 2 diabetes.
The two proteins appear to work synergistically to provide more robust and more permanent weight loss effects.
It is important to note that while preclinical studies suggest promising therapeutic potential, clinical trials in humans are limited.
Further research needs to be done to determine the efficacy and safety profiles.
CJC-Ipamorelin Blend
Blend: CJC IPA 5-5CJC (No Dac) 5 MG. IPAMORELIN 5 MG
CJC-1295 & Ipamorelin Peptide Blend
Overview
The CJC-1295 & Ipamorelin blend combines two extensively studied growth hormone–modulating peptides that act through distinct yet complementary pathways of the growth hormone (GH) axis. Researchers have explored this combination for its potential to support GH pulsatility, tissue repair, metabolic efficiency, and anabolic balance—while maintaining selectivity and physiological feedback control.
Ipamorelin
Ipamorelin is a highly selective growth hormone secretagogue widely studied for its apparent specificity to the growth hormone/ghrelin secretagogue receptor (GHS-R). Unlike many earlier secretagogues, Ipamorelin does not appear to significantly influence the secretion of other pituitary hormones, including prolactin, FSH, LH, TSH, or ACTH. This selectivity has led researchers to classify it as one of the most targeted GH secretagogues identified to date.
Ipamorelin is believed to function through molecular mimicry of ghrelin, a naturally occurring hormone involved in GH release, appetite signaling, and gastrointestinal motility. GH secretion stimulated by Ipamorelin appears to remain regulated by endogenous negative feedback mechanisms, potentially minimizing downstream effects associated with excessive GH exposure.
Based on preclinical observations, researchers have suggested that Ipamorelin may influence multiple ghrelin-associated physiological processes. These include potential roles in:
- Gastrointestinal motility and bowel function
- Muscle development and repair
- Bone growth and remodeling
- Pancreatic insulin signaling
- Cellular repair and collagen synthesis
- Additional hypotheses propose potential involvement in sleep cycle regulation and cognitive function, though these areas remain under continued investigation.
CJC-1295 (Mod GRF 1-29, No DAC)
CJC-1295 (Mod GRF 1-29) is a synthetic analog of somatocrinin, the body’s endogenous growth hormone–releasing hormone (GHRH). Researchers suggest that it binds to and activates the GHRH receptor on pituitary somatotroph cells, promoting pulsatile GH secretion in a manner consistent with physiological rhythms.
Because GH release remains governed by feedback regulation, the peptide appears to support sustained GH signaling without the typical adverse effects associated with continuous overstimulation. Preclinical research has associated CJC-1295 with potential supportive roles in:
- Lean body mass development
- Muscle and bone growth
- Insulin sensitivity
- Cellular repair and regeneration
Animal studies have demonstrated normalization of growth parameters in GHRH-deficient models, including body weight, length, bone dimensions, lean mass, and subcutaneous fat distribution. Additional findings suggest increased pituitary GH mRNA expression and total pituitary RNA content, indicating possible stimulation or proliferation of GH-producing somatotroph cells.
CJC-1295 has also been explored for potential immune-modulating and cognitive support properties.
Mechanistic Insights
At the cellular level, CJC-1295 is thought to initiate GH release by binding to GHRH receptors on pituitary cells, triggering receptor conformational changes and activation of intracellular G-proteins. This process may stimulate second-messenger signaling pathways—such as cAMP and IP₃—leading to protein kinase activation, transcription factor phosphorylation, and increased GH gene expression. The culmination of these events appears to be the fusion of GH-containing vesicles with the cell membrane, enabling hormone release.
Digestive System Research: Ipamorelin
Research investigating Ipamorelin’s effects on gastrointestinal function suggests that activation of ghrelin receptors may accelerate gastric emptying and enhance gastric smooth muscle contractility. In rodent models of postoperative ileus, Ipamorelin administration appeared to significantly reverse surgery-induced delays in gastric emptying and restore contractile responsiveness. These findings have led researchers to hypothesize potential roles in digestive motility support.
Secondary observations in animal models indicated increases in appetite, body weight, fat pad mass, and circulating leptin levels—suggesting altered energy balance and food intake regulation.
CJC-1295 & Ipamorelin Combination
Researchers propose that Ipamorelin may elevate basal GH signaling, while CJC-1295 enhances pulsatile GH release on top of this elevated baseline—supporting both acute and sustained GH activity. Together, the peptides appear to influence the GH axis through complementary mechanisms.
Preclinical studies suggest this combination may support:
- Lean body mass preservation
- Improved nitrogen balance
- Enhanced metabolic efficiency
- Muscle development and recovery
One investigation examining nitrogen metabolism in steroid-induced catabolic states found that Ipamorelin administration was associated with reduced hepatic urea synthesis, downregulation of urea cycle enzymes, restoration of nitrogen balance, and normalization of organ nitrogen content.
Differences in pharmacokinetics may further contribute to synergy: Ipamorelin appears to act rapidly and clear quickly, while CJC-1295 (particularly DAC-bound forms) is known for prolonged GH signaling. Together, this may allow for both rapid onset and sustained GH axis support.
Peptide Specifications
CJC-1295 (Mod GRF 1-29, No DAC)
Molecular Formula: C₁₅₂H₂₅₂N₄₄O₄₂
Molecular Weight: 3367.95 g/mol
Sequence:
H-Tyr-D-Ala-Asp-Ala-Ile-Phe-Thr-Gln-Ser-Tyr-Arg-Lys-Val-Leu-Ala-Gln-Leu-Ser-Ala-Arg-Lys-Leu-Leu-Gln-Asp-Ile-Leu-Ser-Arg-Lys(Mal)-NH₂
Note: Does not contain DAC
Ipamorelin
Molecular Formula: C₃₈H₄₉N₉O₅
Molecular Weight: 711.86 g/mol
Sequence: Aib-His-D-2-Nal-D-Phe-Lys-NH₂
Disclaimer
The products referenced are research chemicals intended solely for laboratory and in-vitro experimentation. They are not intended for human or animal consumption. Any form of bodily introduction is prohibited by law. Sales are restricted to licensed researchers and qualified professionals. All information presented is for educational purposes only.

DSIP 5MG
Delta Sleep-Inducing Peptide (DSIP) 5mg is a synthetic neuropeptide used in research to study the regulation of circadian rhythms, promotion of deep sleep, and potential stress reduction. Often administered via subcutaneous injection, it modulates neurotransmitter activity.
Key Details About DSIP 5mg:
Purpose: Primarily investigated for improving sleep quality, treating insomnia, and potential benefits in pain management and stress reduction.
Administration: Typically administered via subcutaneous (under the skin) injection, though sometimes in studies by IV.
Dosage: Research applications have varied, with some studies focusing on lower doses, making a 5mg vial last for multiple sessions.
Mechanism: It acts on the nervous system, potentially affecting GABA and NMDA neurotransmitter systems to regulate sleep.
Side Effects: Generally well-tolerated, but reported effects can include mild daytime drowsiness, vivid dreams, or rare, mild headaches.
Important Considerations:
DSIP is not FDA-approved for human consumption and is strictly for research purposes. It is a potent regulatory peptide, and its effects on humans are still being studied.
This information is for research purposes only and not medical advice.

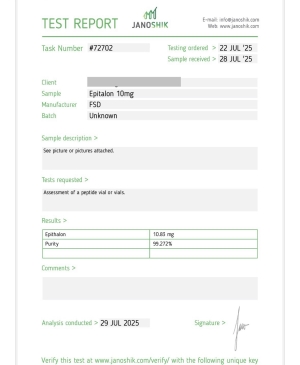
EPITALON 10MG
CAS Number: 307297-39-8
Epitalon (also known as Epithalon or AEDG peptide)
Epitalon – Research Overview
Chemical name: Epithalamin / Epitalon / Epithalon
Sequence: Ala-Glu-Asp-Gly
Class: Synthetic tetrapeptide (4–amino acid peptide)
Primary target: Pineal gland regulatory pathways (hormone-signaling and circadian-rhythm pathways)
Molecular weight: ~390.35 Da
Epitalon is a synthetic peptide modeled after epithalamin, a naturally occurring compound isolated from the pineal gland.
It has been investigated in preclinical research for its effects on:
- Telomere-associated pathways
- Circadian rhythm regulation
- Antioxidant activity
- Stress-response mechanisms
- Cellular aging pathways
All findings refer strictly to animal and cell-based studies, not human clinical outcomes.
Molecular & Pharmacological Profile
In preclinical models, Epitalon has been shown to influence several biological systems connected to aging and cellular regulation.
Research has focused on its potential effects in:
- Telomerase activation (enzyme involved in telomere maintenance)
- Antioxidant pathways (reducing oxidative stress in cells)
- Neuroendocrine signaling (pineal gland activity + melatonin secretion)
- Circadian rhythm modulation (sleep-wake cycle pathways in animal models)
- Immune function support (changes in T-cell activity in rodents)
- These effects are observed only in laboratory environments.
Mechanism of Action (Preclinical)
Epitalon appears to act through several key mechanisms identified in experimental settings:
- Modulation of Telomerase Activity
(increasing activity of the enzyme that preserves telomere length — in vitro findings) - Influence on Melatonin Secretion
(pineal gland signaling that regulates day-night biological rhythms) - Antioxidant Effects
(reduced oxidative stress markers in animal and cell models) - Regulation of Gene Expression Linked to Cellular Stress
(modifying genes related to cell longevity and repair in preclinical studies) - Support of Immune System Parameters in Rodents
(increased lymphocyte activity and improved immune biomarkers in aging animals)
These mechanistic observations make Epitalon useful for research into aging, stress biology, and pineal-gland function.
Preclinical Research: Potential Applications
Epitalon has been studied in several categories of preclinical research, including:
- Aging & Longevity Models
Restoration of telomerase activity (maintaining chromosome stability)
Observed slowing of age-related biomarker decline
Increased life expectancy in certain rodent models (aging-mouse studies) - Circadian Rhythm & Neuroendocrine Pathways
Increased melatonin secretion (sleep-wake hormone)
Resetting of circadian timing in aged rodents
Improved alignment of peripheral clocks in cell models - Antioxidant & Cellular Stress Research
Reduction in reactive oxygen species
Increased cellular resistance to oxidative stress
Upregulation of protective proteins associated with longevity - Immune System Models
Enhanced T-cell activity (improved immune signaling in aging rodents)
Increased overall immune responsiveness in preclinical experiments - Tissue-Repair & Organ-Function Studies
(rodent models only)
Increased regenerative capacity in certain tissues
Improved morphology of aging organs in experimental settings
These applications are observed only in preclinical research, not established therapeutic uses.
Efficacy Observed in Preclinical Models
Longevity Studies (Rodents)
- Epitalon extended average and maximum lifespan in multiple aging-rodent models
- Improved physiological markers associated with aging
- Reduced incidence of age-related pathological changes (organ and tissue level)
Cellular Studies
- Telomerase activation observed in cultured cells (enhanced maintenance of telomere length)
- Reduced DNA damage accumulation under oxidative stress
- Increased expression of protective, pro-repair proteins
Pineal Gland & Circadian Rhythm
- Increased melatonin levels in aging animal models
- Restored circadian rhythm regularity (more stable biological clock patterns)
- Antioxidant Effects
Reduced free radical levels
- Improved mitochondrial function (cells produced energy more efficiently)
All results refer only to animal and in-vitro studies, not human clinical findings.
Safety & Tolerability (Preclinical)
Across preclinical studies:
- Epitalon showed low toxicity at standard research doses
- No evidence of carcinogenic effects in aging-animal studies
- No major adverse reactions reported in rodent models
- Long-term safety, human PK/PD, and clinical tolerability remain unestablished
- Epitalon is not FDA-approved and has not undergone sufficient human research to establish safety.
Regulatory Status & Research Disclaimer
Epitalon is an investigational research peptide.
It is not approved by the FDA or any regulatory agency for therapeutic, dietary, or cosmetic use.
For Research Use Only.
Not for human consumption.
Not for veterinary use.
Not for diagnostic or therapeutic applications.
No medical claims are made or implied.
Researchers are responsible for compliance with all applicable regulations.
Key References (Preclinical)
Anisimov V. et al. Effect of epithalamin on lifespan and aging in rats. Biogerontology.
Khavinson V. et al. Epitalon and telomerase activation in human somatic cells. Bull Exp Biol Med.
Anisimov V. et al. Role of pineal peptides in regulation of aging and tumor development in rodents. Neuro Endocrinol Lett.
Khavinson V., et al. Epitalon: effects on circadian rhythms and endocrine functions. Exp Gerontol.
Linchenko S. et al. Antioxidant properties of pineal-derived peptides in preclinical models. Cell Biochem Funct.
Telomere Elongation: Epitalon is best known for its ability to increase the activity of the enzyme telomerase, which helps to maintain and elongate telomeres, the protective caps on the ends of chromosomes that naturally shorten as cells divide and age. This mechanism is thought to extend the lifespan of cells and delay cellular aging (senescence).
Circadian Rhythm and Sleep Regulation: As a synthetic version of the pineal gland peptide epithalamin, epitalon has been studied for its role in regulating the body's natural sleep-wake cycles (circadian rhythms) by increasing melatonin production. Improved sleep quality is a commonly reported effect in human trials.
Antioxidant Properties: Epitalon exhibits significant antioxidant effects, helping to reduce oxidative stress and the formation of reactive oxygen species (ROS), which can damage cells and contribute to aging and disease.
Cancer and Tumor Suppression: Animal studies have suggested that epitalon may have anti-tumor properties, including reducing the incidence of spontaneous tumors and inhibiting the development of certain types of cancer, such as leukemia. Research indicates it may selectively induce apoptosis (cell death) in cancer cells without harming normal cells.
Immune System Modulation: Studies suggest epitalon can boost immune function by enhancing the production and activity of immune cells (such as T-cells) and regulating inflammatory biomarkers like cytokines.
Cardiovascular Health: Research in elderly patients has suggested that treatment with epitalon's parent extract, epithalamin, can lead to a reduced rate of cardiovascular mortality and improvements in carbohydrate and lipid metabolism, potentially managing conditions like hypertension and atherosclerosis.
Other Potential Benefits: The peptide has also been explored for its potential effects on hormone balance (especially in relation to menopause and reproductive function), neuroprotection, cognitive function, and skin health.
CAS number for Epitalon is 307297-39-8. This number is a unique identifier for the chemical substance, also known by synonyms like Epithalon or Epithalamin, which is a synthetic peptide.
CAS Number: 307297-39-8
Other Identifiers: The CAS number is also linked to other identifiers such as an MDL number (MFCD04034655) and a CID (219042).
Synonyms: Common synonyms include Epithalon, Epithalamin, and its full chemical name, which is based on its amino acid sequence.
Product Note:
All products are shipped in lyphollized or powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.

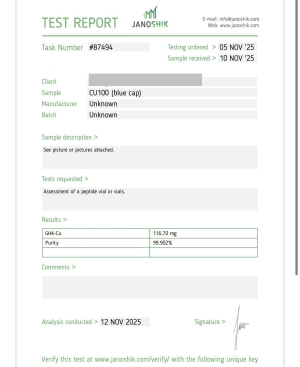
GHK-CU 100MG
GHK-Cu is a synthetic tripeptide bound with copper, presented in lyophilized powder form. This material is provided for research use only and is not intended for human or veterinary applications.
GHK-CU 50MG
Research: Skin/wound repair, hair growth ,Anti-Aging, Nerve regeneration
GHK-Cu – Research Overview
Chemical name: Glycyl-L-histidyl-L-lysine–Copper(II) Complex
Common research name: GHK-Cu
Class: Copper-binding tripeptide
Sequence: Gly-His-Lys
Molecular weight: ~340.8 Da (peptide) / ~403 Da (copper complex)
Primary targets: Wound-healing pathways, extracellular matrix remodeling, anti-inflammatory signaling
GHK-Cu is a naturally occurring tripeptide that forms a complex with copper.
It has been extensively examined in preclinical studies for its effects on:
- Tissue remodeling
- Collagen and ECM production
- Anti-inflammatory signaling
- Hair-follicle biology
- Antioxidant pathways
- Gene-expression regulation
All findings refer to cell-based and animal research, not established effects in humans.
Reddit User Experiences With GHK-Cu
Skin Improvements
Many Reddit users say GHK-Cu noticeably improves skin quality, elasticity, and fine lines.
> “It’s literally erasing the fine lines on my face.”
Another user described thicker, tighter skin after several weeks:
> “The difference is subtle but my skin got thicker and tighter.”
Some users report improvements in skin texture and overall appearance:
> “I saw some plumping and smoother texture after about 8 weeks.”
Hair Thickness and Density
Some Reddit discussions mention thicker hair or improved density.
Example comment:
> “Week 3 and my hair already feels thicker.”
Another user experimenting with hair treatments wrote:
> “It’s been improving overall hair density and thicker strands.”
Anti-Aging and Skin Glow
Several users say GHK-Cu makes their skin look younger or healthier overall.
Example Reddit experience:
> “It was truly remarkable improving the color and texture of my skin.”
Others describe their skin looking “wonderful” after extended use:
> “Skin is wonderful after using it long-term.”
Common Positive Themes From Reddit
Across many threads, people who like GHK-Cu commonly report:
• smoother skin texture
• reduced fine lines
• thicker or firmer skin
• improved hair density
• overall skin “glow”
These experiences align with research showing the copper peptide may stimulate collagen production and improve skin elasticity.
Molecular Pharmacological Profile
GHK-Cu is present naturally in human plasma and tissues.
In preclinical systems, GHK-Cu has been shown to:
- Bind copper ions tightly (stabilizes copper and delivers it into cells)
- Influence MMPs and TIMPs (matrix-degrading enzymes and their regulators)
- Modulate inflammatory cytokines (shift toward lower inflammation)
- Increase collagen, elastin, and glycosaminoglycan production (ECM building blocks)
- Activate repair-associated genes (genes involved in healing and regeneration)
These observations make GHK-Cu a widely studied peptide in research models of tissue repair, skin biology, and regenerative pathways.
Mechanism of Action (Preclinical)
- Copper Delivery to Cells
GHK acts as a copper carrier (delivering copper into cells), which is crucial for enzymes involved in repair and antioxidant defense. - Stimulation of Collagen Remodeling
GHK-Cu increases collagen synthesis and reduces collagen breakdown
(more collagen produced + less destroyed). - Regulation of Matrix Metalloproteinases (MMPs)
GHK-Cu downregulates damaging MMP enzymes and upregulates TIMPs
(shifts tissue from breakdown toward repair). - Anti-inflammatory Signaling
Preclinical models show reduced IL-6, TNF-α, and other inflammatory mediators
(lower cellular inflammation markers). - Enhanced Angiogenesis
GHK-Cu stimulates VEGF and other signals that promote blood vessel formation
(improved oxygen and nutrient delivery in healing models). - Gene Expression Modulation
Genomic analysis shows that GHK-Cu affects hundreds of genes tied to:- Tissue repair
- Antioxidant defense
- Nerve outgrowth
- ECM remodeling
- Anti-aging pathways
(GHK-Cu acts like a “repair switch” at the genetic level in preclinical research.)
Preclinical Research: Potential Applications
GHK-Cu is widely studied in in-vitro and animal models.
- Skin & Tissue Remodeling Research
Studies show GHK-Cu supports:
Increased collagen and elastin (stronger tissue framework)
Improved fibroblast function (cells responsible for building connective tissue)
Enhanced wound matrix remodeling - Anti-inflammatory Research
Reduced inflammatory cytokines
Improved antioxidant enzyme expression
Lowered oxidative stress markers (less cellular damage from ROS) - Hair-Follicle Biology
In rodent and cell studies, GHK-Cu has been associated with:
Increased follicle size
Enhanced follicular signaling pathways
Improved microcirculation (better blood flow around follicles) - Nerve & Tissue Regeneration Models
Increased nerve outgrowth in cell-culture models
Supportive effect on neurotrophic pathways (nerve-survival pathways) - ECM (Extracellular Matrix) Studies
GHK-Cu influences:
- Collagen I & III synthesis
- Glycosaminoglycan production
- Crosslinking and structural integrity of connective tissue
Efficacy Observed in Preclinical Models
- Skin & Connective Tissue
- Up to 70% increase in collagen synthesis in in-vitro fibroblast cultures
- Reduced connective-tissue breakdown enzymes (MMPs)
- Enhanced organization of newly formed tissue
Anti-inflammatory Effects
- Significant reduction in TNF-α, IL-6, and other pro-inflammatory markers
- Increased SOD (superoxide dismutase) and catalase activity (major antioxidant enzymes)
Hair Growth Models
- Improved follicular signaling
- Increased dermal papilla viability (key hair-growth cells)
- Enhanced angiogenesis around hair follicles
Nerve & Organ Models
- Increased neurite outgrowth (longer nerve branches)
- Better cellular viability under stress
- Upregulated genes tied to regeneration and survival
Safety Tolerability (Preclinical)
- GHK-Cu shows low toxicity in preclinical models
- No evidence of mutagenic or carcinogenic activity in available data
- Copper-binding reduces free-copper toxicity
Long-term human safety, dosing, and PK/PD remain unestablished
GHK-Cu is not FDA-approved for medical use.
Regulatory Status
GHK-Cu is an investigational research compound.
It is not approved by the FDA or any regulatory body for therapeutic, dietary, or cosmetic use.
For Research Use Only.
Not for human consumption.
Not for veterinary use.
Not for diagnostic or therapeutic applications.
Key References (Preclinical)
Pickart L. The human tripeptide GHK and tissue remodeling. J Biomater Sci Polym Ed.
Maquart F.X. et al. Stimulation of collagen synthesis by GHK-Cu in fibroblast cultures. Cell Mol Biol.
Pickart & Margolina. GHK-Cu as a gene-modulating peptide. BioMed Research Int.
Siméon A. et al. GHK-Cu and MMP/TIMP modulation in connective tissue. Arch Dermatol Res.
Canapp S. Copper peptide–mediated wound healing in animal models. Vet Surg.
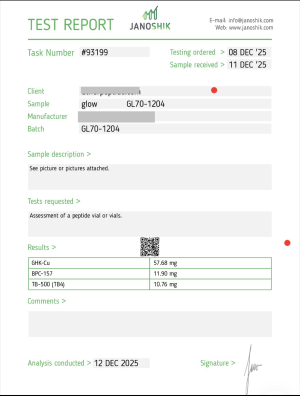
GLOW 70
Blend (GHK-Cu, BPC-157, TB-500) 70mg
This research peptide blend combines three regenerative peptides into a single vial for studies examining complementary tissue repair pathways.
GHK-Cu up-regulates wound healing processes and drives collagen production, elastin, and angiogenic growth-factor expression in laboratory models.
BPC-157 exhibits gastro-protective, soft-tissue repair, and anti-inflammatory actions through nitric-oxide signaling, growth-factor receptor modulation, and cytokine balance.
TB-500 (Thymosin Beta-4 Fragment) enhances cell migration and angiogenesis via actin-sequestering and integrin-linked pathways.
Researchers can examine potential synergy across copper-mediated extracellular-matrix activation (GHK-Cu), cytoprotective signaling (BPC-157), and actin-dependent cell motility (TB-500). In vitro and ex vivo models evaluate collagen deposition rates, angiogenic indices, and recovery metrics following controlled tissue injury.
Composition: 70 mg lyophilized blend per vial
50 mg GHK-Cu | 10 mg BPC-157 | 10 mg TB-500

Glutathione 600MG
Glutathione Injection – 200mg/600mg/1500mg (Research Use Only)
Cellular Defense • Detox Support • Oxidative Stress Reduction
Overview
Glutathione is the body’s master antioxidant — a tripeptide found naturally in every cell, responsible for neutralizing free radicals, supporting detoxification pathways, and maintaining overall cellular resilience. In research environments, glutathione is commonly studied for its role in redox balance, mitochondrial protection, and immune modulation.
Research Highlights
Powerful Antioxidant Activity
Glutathione participates in redox cycling, directly neutralizing reactive oxygen species (ROS) and reducing oxidative stress in test subjects.
Detoxification Pathway Support
A key cofactor in Phase II liver detoxification. Studies show enhanced breakdown and elimination of toxins and metabolic byproducts.
Mitochondrial Function & Energy Support
Assists with maintaining mitochondrial membrane integrity, preserving cellular energy output in research models.
Skin Brightening & Pigmentation Modulation
Frequently studied for modulation of melanogenesis — resulting in a brighter, more even appearance in certain test scenarios.
Immune Regulation
Plays a central role in lymphocyte proliferation, inflammatory cytokine reduction, and general immune homeostasis.
Aging & Cellular Protection
Glutathione helps slow oxidative aging processes by protecting lipids, proteins, and DNA from damage.
Why Researchers Use Glutathione
Hospira Bac Water 30 ML
Bacteriostatic Water 30ML is a sterile laboratory solution formulated with benzyl alcohol for use in controlled research environments. Researchers use this solution to support preparation and dilution workflows that require consistent sterile handling.
Sterile bacteriostatic water formulated with benzyl alcohol
Used to support controlled preparation and dilution in research settings
Designed for consistent handling across laboratory workflows

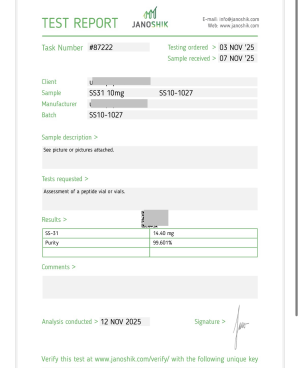
Ipamorelin 10
Ipamorelin (10 MG)
Ipamorelin – Research Overview
Chemical name: Aib-His-D-2-Nal-D-Phe-Lys-NH₂
Class: Selective growth hormone secretagogue (GHS)
Primary target: Ghrelin / Growth Hormone Secretagogue Receptor (GHSR-1a)
Molecular weight: ~711.9 Da
Activity: Mimics the action of ghrelin on pituitary growth hormone release (signals GH secretion without affecting other hormones)
Ipamorelin is a synthetic pentapeptide investigated in preclinical research for its highly selective ability to stimulate growth hormone release (GH secretion) through the ghrelin receptor.
It is known for its selectivity, meaning it does not significantly alter cortisol, prolactin, or ACTH in research models
(unlike some older GHS peptides).
Molecular & Pharmacological Profile
In preclinical models, Ipamorelin has been shown to:
- Activate GHSR-1a receptors (growth hormone secretagogue receptor)
- Increase pulsatile GH release (natural GH spikes)
- Avoid significant activation of other pituitary hormones
- Display high specificity to ghrelin pathways (minimal off-target effects)
- Produce dose-dependent GH secretion in animal models
These characteristics make Ipamorelin a widely used peptide in endocrine and metabolic research.
Mechanism of Action (Preclinical)
Ipamorelin acts through the ghrelin receptor, producing GH release in a controlled and predictable way.
- Binding to GHSR-1a (ghrelin receptor)
(the receptor ghrelin uses to stimulate GH secretion) - Triggering GH release from the anterior pituitary
(increases natural GH pulses in preclinical models) - Lower effect on ACTH and cortisol pathways
(reduced side-hormone stimulation compared to GHRP-6 or Hexarelin) - Mimicking natural GH rhythms
(GH rises in pulses rather than a constant elevation) - Minimal impact on appetite signaling
(preclinical studies show weaker hunger stimulation than true ghrelin)
Preclinical Research: Potential Applications
Ipamorelin has been investigated across various animal and cell-based research contexts:
- Growth Hormone & Endocrine Research
Study of GH release mechanisms
Investigation into GHSR-1a pathway signaling
Analysis of pulsatile GH vs. continuous GH patterns - Metabolic Research
Interactions between GH and fat metabolism
GH-induced effects on energy expenditure
Observing GH’s impact on body composition models
(GH affects fat and lean-mass pathways in animals) - Bone & Connective Tissue Research
GH-stimulated IGF-1 expression in tissues (downstream GH effects)
Preclinical studies on bone remodeling markers
Exploring GH’s role in collagen synthesis - Neurological & Cognitive Models
GH pathways have been connected to:
Neural plasticity
Cognitive performance
Neuroprotection mechanisms
(these findings apply to GH pathways, not directly to human outcomes) - Anti-inflammatory & Recovery Pathways (Preclinical)
GH upregulates:
- Repair-associated genes
- Anti-inflammatory pathways
- Cellular regeneration markers
- Ipamorelin is often studied alongside CJC-1295 for combined GH modulation, but this overview focuses on Ipamorelin alone.
- Efficacy Observed in Preclinical Models
- Growth Hormone Release
- Strong, dose-dependent GH pulse increases
- Less desensitization vs. older GHRPs
- Minimal stimulation of non-GH pituitary hormones
(very low prolactin and ACTH activation)
Metabolic Findings
Rodent studies show:
- Increased lipolysis (breaking down stored fat)
- Increased fatty-acid oxidation (burning fat for energy)
- Improved metabolic efficiency in GH-regulated pathways
Bone and Tissue Studies
GH-related findings include:
- Increased collagen synthesis
- Enhanced osteoblast activity (bone-building cells)
- Upregulation of IGF-1 in certain tissues
Neurological Findings
GH modulates:
- Neuronal survival pathways
- Synaptic plasticity markers
Learning and memory biomarkers
(Ipamorelin’s effects here are indirect through GH pathways.)
Key areas of study and potential use include:
Growth Hormone Deficiency (GHD) treatment: As it stimulates GH release, it has been investigated as a potential treatment for both adult and childhood GHD.
Body Composition Improvement: Studies show potential for increasing lean muscle mass and reducing body fat, making it a focus in age management and fitness contexts.
Bone Health: Research, primarily in animal models, has indicated that ipamorelin may increase bone mineral density and enhance bone formation, suggesting potential in the prevention and treatment of osteoporosis.
Wound and Injury Healing: By promoting cellular repair and collagen production, it is being investigated for enhancing recovery from injuries, exercise, or surgery.
Sleep Quality: Clinical and preclinical evidence suggests ipamorelin can improve sleep architecture, specifically enhancing slow-wave sleep duration and overall sleep quality.
Metabolic Syndrome and Type 2 Diabetes Management: It is being investigated for its potential to improve insulin sensitivity and lipid profiles.
Anti-Aging and Regenerative Medicine: Due to the natural decline of GH with age, ipamorelin is used in age management protocols to help mitigate related symptoms like fatigue, decreased muscle tone, and reduced skin elasticity.
Neuroprotection and Cognitive Function: Emerging studies suggest potential benefits for cognitive function, including memory and focus, and neuroprotective effects.
HIV-associated Wasting: Ipamorelin has been an investigational use for managing muscle atrophy associated with conditions like HIV.
Safety & Tolerability (Preclinical)
Ipamorelin is considered one of the most selective GH secretagogues in preclinical research:
-
Low off-target hormone activation
(minimal cortisol, prolactin, and ACTH changes) -
Well-tolerated in rodent studies at standard research doses
-
Low desensitization rate
(receptor continues responding to stimulation) -
No significant effect on hunger
(unlike ghrelin mimetics such as GHRP-6) -
Long-term human safety remains undetermined, and Ipamorelin is not approved for therapeutic use.
Regulatory Status & Disclaimer
- Ipamorelin is an investigational research peptide.
- It is not approved by the FDA or any regulatory body for medical, dietary, cosmetic, or therapeutic use.
- For Research Use Only.
- Not for human consumption.
- Not for veterinary use.
- Not for diagnostic or therapeutic purposes.
- No medical claims are made or implied.
Key References (Preclinical)
Raun K. et al. Ipamorelin: Selective GH secretagogue profile in rats. Endocrinology.
Svensson J. et al. GHSR-1a pathway activation by Ipamorelin. Growth Horm IGF Res.
Broglio F. et al. Comparison of Ipamorelin with GHRP-6 in endocrine models. J Clin Endocrinol Metab.
Kojima et al. Ghrelin receptor signaling pathways and GH release. Nature.
Hickey G. GH secretagogues and metabolic modulation in animal models. Peptides.
Product Note:
All products are shipped in lyphollized or powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.
Storage: Peptides should be stored in a dry, cool, dark place. For best preservation, store at 4°C or colder away from bright light.
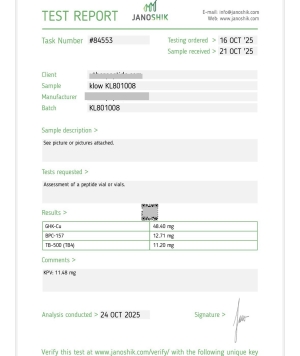
KLOW 80 MG
Blend: GHK-Cu, TB-500, BPC-157, and KPV
The KLOW 80 MG Peptide Blend combines GHK-Cu, TB-500, BPC-157, and KPV to support tissue regeneration, wound healing, and inflammation reduction in non-human test subjects, enhancing recovery and overall wellness in controlled laboratory research environments.
KLOW Blend – Research Overview
Components:
GHK-Cu – copper-binding tripeptide
TB-500 – actin-modulating peptide fragment
BPC-157 – cytoprotective gastric peptide
KPV – α-MSH–derived anti-inflammatory tripeptide
Class: Multi-pathway regenerative research blend
Research Focus: Tissue remodeling, angiogenesis, cellular migration, inflammation modulation, and epithelial/skin repair models.
The KLOW Blend combines four synergistic peptides widely studied in preclinical models for their complementary roles in cellular repair, inflammation signaling, and tissue structural organization.
All findings refer strictly to animal and cell-based research, not established human outcomes.
Component Breakdowns (Preclinical)
-
GHK-Cu (Copper Tripeptide)
Key Research Pathways- Increases collagen, elastin, and glycosaminoglycans (connective tissue components)
- Strong antioxidant effects
- Modulates hundreds of repair-related genes
- Supports angiogenesis in cell models (the formation of new blood vessels)
- Reduces inflammatory markers
- GHK-Cu is often studied for skin and connective-tissue remodeling due to its ECM (extracellular matrix) effects.
-
TB-500 (Thymosin Beta-4 Fragment)
Key Research Pathways- Enhances actin polymerization (crucial for cell movement)
- Increases cell migration
- Supports angiogenesis
- Improves structural repair in muscle/tendon models
- Anti-inflammatory signaling
*TB-500 is a powerful mobilizer of repair cells, moving them toward injury sites in rodent studies.
-
BPC-157
Key Research Pathways- Cytoprotection of GI, liver, vascular tissue
- Increased fibroblast recruitment (cells that build new tissue)
- Stabilization of vascular endothelium (The thin layer of cells that lines the inside of all your blood vessels. Think of the endothelium as:
“The protective, intelligent lining that keeps blood vessels healthy and functioning smoothly.”) - Strong angiogenic activity
- Reduced oxidative stress
- BPC-157 is widely researched for multi-tissue regenerative signaling.
-
KPV (Lys-Pro-Val)
A potent anti-inflammatory tripeptide derived from α-MSH.
Key Research Pathways- Downregulates TNF-α, IL-6, NF-κB
- Supports epithelial barrier integrity (The cells that make up the outer covering or lining of surfaces in the body.)
- Reduces inflammatory cell infiltration
- Improves wound environment in rodent models
- KPV is a pure anti-inflammatory signaler, complementing the structural/repair focus of the other three.
Synergistic Mechanisms (Preclinical)
The KLOW Blend addresses all phases of tissue restoration:
-
Inflammation Reduction → (KPV + BPC-157)
- Lowered cytokines
- Reduced local inflammation
- Stabilized endothelial and epithelial barriers
(Creates a “calmer” environment for repair.)
-
Cell Migration Support → (TB-500)
- Actin remodeling
- Faster movement of repair cells
- Increased angiogenic signaling
(Helps deliver cells to where they are needed.)
-
Tissue Remodeling → (GHK-Cu + BPC-157)
- Collagen production
- Antioxidant protection
- Improved ECM structure
- Enhanced organization of new tissue
(Supports the rebuilding phase.)
-
Angiogenesis → (TB-500 + BPC-157 + GHK-Cu)
- Increased capillary formation
- Better nutrient/oxygen delivery
- Improved microcirculation
(Strong multi-peptide vascular synergy.)
Preclinical Research: Potential Applications
The KLOW blend is typically studied in:
-
Musculoskeletal Research
- Tendon and ligament repair markers
- Muscle-fiber regeneration
- Collagen deposition
- Reduced inflammatory fibrosis
- Improved mobility and structural integrity in rodent models
-
Skin & Dermal Biology
Increased collagen Type I & III- Improved wound closure
- Reduced redness and inflammatory cell infiltration
- Enhanced keratinocyte migration
- GHK-Cu + KPV is a common pairing in dermal research.
-
Vascular & Endothelial Studies
- Protection against oxidative stress
- Repair of endothelial lining
- Better angiogenic responses
-
GI & Mucosal Models
(BPC-157 + KPV dominate these pathways)- Reduced inflammatory damage
- Improved epithelial tight-junction formation
- Faster mucosal repair in rodent models
-
Systemic Inflammatory Models
- Decreased TNF-α and IL-6 (pro-inflammatory cytokines
(→ chemical signals that trigger inflammation in the body). - Reduced ROS and oxidative damage
- Excess ROS can cause:
oxidative stress
cellular damage
accelerated tissue aging
inflammation - Normalized immune response patterns
- Decreased TNF-α and IL-6 (pro-inflammatory cytokines
Efficacy Observed in Preclinical Studies
Inflammation
- Significant cytokine reduction
- (Cytokines are small messenger proteins that cells release to communicate with each other during:
inflammation,immune responses, healing,stress, injury
Think of them as the “text messages” of the immune system.
They tell cells: when to increase/decrease inflammation, recruit immune cells,repair tissue,fight infection - Less local tissue swelling
- Reduced inflammatory infiltrates
- Repair
- Faster wound closure
- Stronger collagen alignment
- Greater fibroblast density (fibroblast=cells that build and repair connective tissue:collagen, tendons, ligaments, skin, fascia. So more connective-tissue repair cells in an area (the cells that produce collagen).
- Increased angiogenesis
Cellular Protection
- (Cytokines are small messenger proteins that cells release to communicate with each other during:
- Lower oxidative stress
- Improved tissue preservation
- Enhanced mitochondrial stability
All findings are animal/cell-based, not confirmed in humans.
Safety & Tolerability (Preclinical)
Across available studies:
- All four peptides demonstrate low toxicity
- No major organ-damage signals at standard research doses
- Good tolerability
- No hormonal disruption
- Long-term human PK/PD unknown
- None of these compounds are FDA-approved.
Regulatory Status & Disclaimer
- The KLOW blend is an investigational research formulation.
- None of the components are approved by the FDA for therapeutic, dietary, or cosmetic use.
- For Research Use Only.
- Not for human consumption.
- Not for veterinary use.
- Not for diagnostic or therapeutic applications.
- No medical claims are made or implied.
- Researchers must follow all applicable laws and regulations.

Lipo-B 216MG
Research: liver health, energy, mental clarity
Methionine: An essential amino acid that helps the liver process fatty acids, lowers cholesterol, and prevents the buildup of excess fat in the liver and arteries.
Inositol: A B-vitamin related compound that aids in fat and cholesterol metabolism and supports optimal brain function.
Choline: An essential nutrient vital for liver health and fat metabolism, which helps transport and excrete fats.
Vitamin B12 (Cyanocobalamin/Methylcobalamin): Essential for energy production, red blood cell formation, and overall metabolic processes.
B vitamins (like B1, B2, B6)
L-carnitine.
Lipo-B injections are used to research:
Support weight loss efforts: enhance the subject's ability to break down fat and may help test subjects break through weight loss plateaus, but are not a standalone solution and must be combined with diet and exercise.
Increase energy levels: The Vitamin B12 component helps convert food into energy, which can combat fatigue often associated with dieting.
Improve liver function: The lipotropic agents aid the liver in detoxification and processing fats more efficiently.
Enhance mood and mental clarity: B vitamins are known to support nervous system function, potentially improving focus and mood.

Lipo-C 526MG
Fat Blaster
L-Carnitine 20 mg
L-Arginine 20 mg
Methionine 25 mg
Inositol 50 mg
Choline 50 mg
B6 (Pyridoxine) 25 mg
B5 (Dexpanthenol) 25 mg
B12 (Methylcobalamin) 1 mg
A high‑potency blend of lipotropic nutrients, vitamin C, and B‑complex vitamins used in research for liver disease; increases liver enzymes; and improves fat metabolism and energy.
All products are shipped in lyphollized or powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.

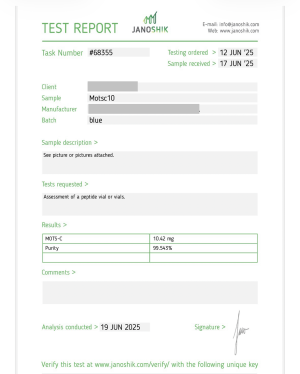
Mots-c 10MG
CAS Number: 1627580-64-6
MOTS-C acronym stands for "mitochondrial-derived peptide" and it is also known by its CAS number, 1627580-64-6. MOTS-c is studied for the role it plays in regulating energy metabolism, stress, and aging. It is produced in response to exercise or stress and functions as a type of "mitochondrial hormone" or "mitokine".
What it is: A bioactive peptide encoded by the mitochondrial 12S rRNA gene.
Function: It promotes energy metabolism and stress homeostasis, with potential roles in improving insulin sensitivity, exercise capacity, and longevity.
MOTS-c – Research Overview
Full name: Mitochondrial Open Reading Frame of the 12S rRNA type-c
Class: Mitochondrial-derived peptide (MDP)
Sequence: 16–amino acid peptide
Primary target: AMPK activation + mitochondrial stress-response pathways
Molecular weight: ~2 kDa
MOTS-c is a peptide encoded within mitochondrial DNA (not nuclear DNA), making it part of a unique class of molecules known as mitochondrial-derived peptides (MDPs).
It has been investigated in preclinical studies for its effects on:
- Metabolic regulation
- Cellular energy production
- Stress-response pathways
- Insulin signaling
- Mitochondrial function
All findings refer to animal and in-vitro research, not established human outcomes.
Molecular & Pharmacological Profile
In research models, MOTS-c functions as a mitochondrial stress signal that helps regulate:
- Energy expenditure
- Glucose use
- Fat metabolism
- Cellular homeostasis (cell balance and stability)
MOTS-c is known to:
- Activate AMPK (energy sensor that increases fat burning + glucose uptake)
- Improve mitochondrial efficiency (better use of fuel inside the cell)
- Regulate nuclear gene expression (signals from mitochondria to nucleus)
- Enhance cellular stress tolerance (more resilient cells during metabolic stress)
These features make it valuable in metabolic and aging-related research models.
Mechanism of Action (Preclinical)
- Activation of AMPK (energy-sensing enzyme)
AMPK increases:- Fatty-acid oxidation (burning fat for energy)
- Glucose uptake (cells use more sugar for fuel)
- Cellular resilience during stress
- Regulation of Nuclear Genes
MOTS-c triggers a process called mitonuclear communication
(mitochondria sending signals to the nucleus to change gene expression).
This affects:- Metabolism
- Stress response
- Repair pathways
- Improved Mitochondrial Function
Preclinical models show:- More efficient ATP production (cellular energy)
- Greater mitochondrial density in muscle tissue
- Enhanced resistance to metabolic damage
- Increased Fat Burning
Via AMPK and mitochondrial pathways, MOTS-c enhances:
Fatty-acid oxidation (fat → energy)
Use of fat stores during activity - Enhanced Cellular Stress Tolerance
Research demonstrates improved survival and function in cells exposed to:- Oxidative stress
- Metabolic stress
- High-fat diets
- Aging models
Preclinical Research: Potential Applications
MOTS-c has been studied extensively in rodent and cell-based models:
- Metabolic Regulation Models
- Increased insulin sensitivity (cells respond better to insulin)
- Improved glucose utilization
- Reduced fat accumulation in high-fat diet rodents
- Enhanced AMPK-driven energy pathways
- Obesity & Body-Weight Research
Rodent studies show:- Reduced weight gain
- Increased energy expenditure
- Higher fat oxidation
- Improved metabolic flexibility (cells switching between fuels more easily)
- Exercise & Muscle Performance Models
MOTS-c has been linked to:- Increased muscle endurance
- Better glucose uptake in muscle cells
- Enhanced mitochondrial biogenesis (more / stronger mitochondria)
- Cellular Stress & Longevity Pathways
- Greater resistance to oxidative damage
- Upregulation of protective genes
- Improved metabolic resilience in aging models
- Inflammatory & Immune Pathways (Preclinical)
- Reduced inflammatory cytokines
- Mild immunomodulatory effects
(shifts in immune markers in mice under metabolic stress)
Efficacy Observed in Preclinical Models
Rodent Metabolic Studies
-AMPK activation led to increased fat burning
-Improved insulin signaling
-Reduced adiposity (less stored fat)
-Enhanced physical endurance
-Protection against diet-induced obesity
Cellular Models
-Increased mitochondrial efficiency
-Greater energy availability (ATP)
-Reduced oxidative stress
-Better cell survival under stress conditions
Aging Models
-Improved metabolic biomarkers in older rodents
-Enhanced mitochondrial gene expression
-Greater exercise capacity
All findings are limited to preclinical research, not documented human effects.
Safety & Tolerability (Preclinical)
- No major toxicity reported in rodent studies
- Demonstrated good tolerability at research doses
- Long-term safety, pharmacokinetics, and human interactions remain unestablished
- MOTS-c is not FDA-approved
Regulatory Status & Disclaimer
- MOTS-c is an investigational research peptide.
- It is not approved for therapeutic, dietary, cosmetic, or medical use.
- For Research Use Only.
- Not for human consumption.
- Not for veterinary use.
- Not for diagnostic or therapeutic purposes.
- No medical claims are made or implied.
Key References (Preclinical)
Lee C. et al. MOTS-c as a mitochondrial-derived peptide regulating metabolism. Cell Metabolism.
Reynolds J. et al. AMPK activation by MOTS-c in high-fat diet animals. Aging Cell.
Kim S.J. et al. Role of MOTS-c in exercise and mitochondrial biogenesis. Sci Rep.
Fuku N. et al. Mitochondrial peptides and metabolic stress response. J Physiol.
Yang C. et al. MOTS-c and mitonuclear communication pathways. PNAS.
Product Care:
Store in a cool, dry place away from light. If Constituted, Please Refrigerate. For longer term storage, freezing at -20°C is recommended to maintain integrity.
Product Note:
All products are shipped in lyphollized or powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.

MT-1 10 MG
CAS Number: 75921-69-6
MT1 (Melanotan I) – 10mg Research Peptide
Category: Research Peptides
Form: Lyophilized Powder
Purity: High (research-grade)
Overview
MT1 (Melanotan I) is a peptide studied for its interaction with melanocortin receptors, particularly those involved in pigmentation pathways.
Compared to similar compounds, MT1 is often explored for its more gradual and controlled response, making it of interest in research focused on steady melanin production and longer-term outcomes.
Research Highlights
Studied for support of melanin production pathways
Explored for gradual pigmentation response
Investigated for interaction with MC1 receptors
Often compared to MT2 for a more stable, less aggressive profile
Why Researchers Choose MT1
- Slower, more controlled progression
- Often described as more predictable over time
- Lower incidence of intense short-term effects (anecdotally)
- Suitable for longer observation periods
Common Research Protocols (for informational purposes only)
- Typically begins with low microgram dosing
- Gradual titration over time
- Often used in consistent daily or near-daily schedules
- Research cycles may extend several weeks to months
(Protocol varies depending on research goals.)
Storage & Handling
- Store in a cool, dark place before reconstitution
- Refrigerate after mixing
- Use bacteriostatic water in research settings
- Avoid heat and repeated temperature changes
What You’ll Receive
1 x MT1 (Melanotan I) vial – 10mg lyophilized powder
Secure packaging
GreyVita quality assurance
⭐ What Researchers Are Saying (Reddit Feedback)
⭐ “The response was slower than MT2, but way more even and controlled. I actually prefer it.”
— u/steadyresults • r/Peptides
⭐ “MT1 feels more predictable. It takes patience, but the results build nicely over time.”
— u/longgamebio • r/ResearchChemicals
⭐ “Definitely a ‘slow and steady’ compound. Once it starts working, it’s very consistent.”
— u/gradualgains • r/Peptides
⭐ “I didn’t get the strong side effects like I did with MT2. Much smoother overall experience.”
— u/labnotesdaily • r/PeptideSource
⭐ “Took longer to notice, but it looked more natural in the end.”
— u/cleanprotocol • r/Biohackers
Common Themes from Community Feedback
Gradual, steady progression
More controlled vs aggressive response
Often described as “smoother” than MT2
Requires patience, but builds over time
⚠️ Important Notice
This product is intended for research and laboratory use only.
Not for human consumption.
This product is not intended to diagnose, treat, cure, or prevent any disease.
All information provided is for educational and informational purposes only.
Why GreyVita
Clean, professional presentation
Reliable sourcing
Straightforward, no-hype information
Designed for serious research use

MT-2 10MG
CAS Number: 121062-08-6
Melanotan II (MT-2) is a synthetic peptide analog of the naturally occurring alpha-melanocyte-stimulating hormone (α ????-MSH). It is primarily known for its ability to stimulate melanin production, leading to skin tanning without significant UV exposure. While popular in the bodybuilding and aesthetic communities for tanning, it also acts on other melanocortin receptors, resulting in additional effects on appetite and sexual function.
What Researchers Are Saying (Reddit Feedback)
⭐ “Started low and worked my way up—noticed effects way quicker than I expected. Definitely one of the more interesting peptides I’ve researched.”
— u/peptideexplorer • r/Peptides
⭐ “MT2 has been one of the most consistent compounds I’ve worked with. Easy to manage once you dial in your protocol.”
— u/labratlife • r/FitnessPeptides
⭐ “The response was gradual but very noticeable over a few weeks. Less is definitely more with this one.”
— u/researchmode • r/ResearchChemicals
☀️ Tanning Effects
“One vial lasts me months of maintaining a light, even, natural-looking tan.”
“You get darker so your muscles look more defined haha.”
????️ Appetite Suppression / Weight Effects
“It seems to really curb my appetite.”
“I’m also starting to experience lost appetite.”
“Bad nausea… loss of appetite… seems to be somewhat common.”
???? Sexual / Libido Effects
“Plus the libido spike…”
Key Benefits and Effects
Sunless Tanning: The primary use of Melanotan II is to induce a deep, long-lasting tan by increasing melanin production in the skin, which can occur even with minimal UV exposure.
UV Protection: By boosting melanin, it may offer increased natural protection against UV rays, potentially reducing the risk of sunburn for fair-skinned individuals.
Appetite Suppression: It is known to act on the central nervous system to suppress appetite and, in some cases, aid in weight management.
Enhanced Libido and Sexual Function: Melanotan II can boost libido and improve erectile function in men (and sometimes women) due to its action on the central melanocortin receptors.
Potential Therapeutic Uses: Research has suggested potential benefits for treating conditions like erectile dysfunction and, in some contexts, offering neuroprotective effects.
Important Safety Information and Risks
Melanotan II is not approved by the FDA
Important Notice
This product is intended for research and laboratory use only.
Not for human consumption.
Not intended to diagnose, treat, cure, or prevent any disease.

NAD+ 500MG
CAS Number: 53-84-9
Product Name: NAD+
500 MG
CAS Number: 53-84-9
Molecular Formula: C21H27N7O14P2
Molecular Weight: 663.43 g/mol
Nicotinamide Adenine Dinucleotide
NAD+ is a coenzyme studied for its role in energy metabolism and cellular function. As part of the Electron Transport Chain, it carries electrons that drive ATP production and redox balance. Stored properly, NAD+ 500MG provides researchers with a consistent tool for exploring mitochondrial activity, energy regulation, and cellular pathways.
KEY BENEFITS OF NAD+
Supports research into cellular energy metabolism
Provides insights into mitochondrial function
Acts as an electron carrier in the Electron Transport Chain
Helps investigate redox balance in cells
Offers a stable and consistent format for laboratory use
Useful in studies of energy regulation and cellular pathways
Trusted tool for controlled biochemical and molecular research
Nicotinamide Adenine Dinucleotide, or NAD+, is the oxidized form of NADH. This coenzyme plays a central role in how cells make energy. As part of the Electron Transport Chain, NAD+ carries electrons from biological reactions. Because of this role, it is essential for research on energy metabolism and redox balance.
Researchers use NAD+ 500MG to study how cellular energy links to mitochondrial activity and other pathways. In addition, it supports work that explores how cells adapt and stay in balance. Therefore, NAD+ 500MG serves as a dependable compound for many laboratory studies on cell health.
Each vial contains lyophilized powder that keeps the peptide stable during storage. Researchers dissolve it into a solution before use. For best care, store it in a cool, dry place away from light. After reconstitution, refrigerate it right away. For long-term storage, freeze it at –20°C to protect its integrity.
All products meet pharmaceutical-grade standards but exist only for research and development. They are not for human or animal use, and no dosing details are supplied. In conclusion, NAD+ 500MG remains a trusted tool for studying energy, mitochondria, and cellular function.

PT-141 10MG
PT-141 is a synthetic melanocortin receptor agonist structurally derived from the analog Melanotan II. It binds selectively to MC3R and MC4R, modulating neuropeptide signaling pathways involved in hypothalamic control mechanisms. PT-141 is commonly employed in research focusing on GPCR activation, neuronal signaling, and neuroendocrine modulation.
Researched benefits: Increases sexual arousal Improved erectile function Helps with HSDD Improves sexual pleasure Improves blood pressure Appetite regulation.
Many Reddit users say PT-141 strongly increases libido and arousal, often more than drugs like Viagra because it acts in the brain rather than just improving blood flow.
Example comment:
“PT-141 feels completely different… rock hard erections and increased pleasure.”
Another user described the sensation as heightened overall arousal: “Everything feels richer… faint sensations become pleasurable.”
Some users report very strong erections and increased sexual stamina: “With PT-141 I had incredible erections. My wife loved it.”
Others say it works for both men and women: “It’s marketed as ‘female Viagra’ but it most certainly works for men.”
Common positive themes reported on Reddit: • stronger libido (“puts you in the mood”) • easier erections • heightened physical sensations • longer sexual sessions Many users also say the effect lasts much longer than Viagra, often 10–24 hours after dosing.
For research use only. Not for human consumption.
Contraindications: Not for women who have gone through menopause or for use by men,
RETA 10MG
CAS Number: 2381089-83-2
R10 10MG is a GGG tri-agonist that targets GLP-1 and GIP receptors, akin to standard GLP-1 agonists, but also uniquely activates the glucagon receptor.
Reddit User Experiences With Retatrutide
Many Reddit users report extremely fast weight loss and strong appetite suppression.
Example user comment:
“I started at 350 lbs… now 271 lbs in about 4½ months.”
Another user described dramatic weight loss early in treatment:
“In the first 2 months, I lost 19 kg (about 42 lbs).”
Some people say it significantly improved metabolic health markers during clinical trials:
“BMI went from 34.7 to 23.2… lost 74 lbs.”
Users often mention these benefits:
• strong appetite suppression
• reduced “food noise”
• rapid fat loss
Clinical Data:
GLP-R10: Triple–Hormone-Receptor Agonist for Obesity — A Phase 2 Trial
GLP-R10: A Study (LY3437943) in Participants With Obesity and Cardiovascular Disease (TRIUMPH-3)
GLP-R10: Triple-Hormone-Receptor Agonist Study for Obesity – A Phase 2 Trial
Product Note:
All products are shipped in lyphollized or powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.
Product Name: Reta
CAS: 2381089-83-2
Molecular Mass: 4731.33 g/mol
Molecular Formula: C223H343F3N46O70
PubChem ID: n/a
Storage: Peptides should be stored in a dry, cool, dark place. For best preservation, store at 4°C or colder away from bright light.
RETA 15MG
CAS: 2381089-83-2
R 15MG is a GGG tri-agonist that targets GLP-1 and GIP receptors, akin to standard GLP-1 agonists, but also uniquely activates the glucagon receptor.
Reddit User Experiences With Retatrutide
Many Reddit users report extremely fast weight loss and strong appetite suppression.
Example user comment:
“I started at 350 lbs… now 271 lbs in about 4½ months.”
Another user described dramatic weight loss early in treatment:
“In the first 2 months, I lost 19 kg (about 42 lbs).”
Some people say it significantly improved metabolic health markers during clinical trials:
“BMI went from 34.7 to 23.2… lost 74 lbs.”
Users often mention these benefits:
• strong appetite suppression
• reduced “food noise”
• rapid fat loss
Product Note:
All products are shipped in lyphollized or powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.
Product Name: Reta
CAS: 2381089-83-2
Molecular Mass: 4731.33 g/mol
Molecular Formula: C223H343F3N46O70
PubChem ID: n/a
Storage: Peptides should be stored in a dry, cool, dark place. For best preservation, store at 4°C or colder away from bright light.

Reta 20MG
CAS Number: 2381089-83-2
Retatrutide (Research Peptide)
Retatrutide is a next-generation investigational peptide studied for its unique triple-agonist activity across the GLP-1, GIP, and glucagon receptors. This multi-pathway mechanism has made retatrutide a subject of growing interest in metabolic and energy-regulation research.
Reddit User Experiences With Retatrutide
Many Reddit users report extremely fast weight loss and strong appetite suppression.
Example user comment:
“I started at 350 lbs… now 271 lbs in about 4½ months.”
Another user described dramatic weight loss early in treatment:
“In the first 2 months, I lost 19 kg (about 42 lbs).”
Some people say it significantly improved metabolic health markers during clinical trials:
“BMI went from 34.7 to 23.2… lost 74 lbs.”
Users often mention these benefits:
• strong appetite suppression
• reduced “food noise”
• rapid fat loss
In preclinical and early clinical research settings, retatrutide has been explored for its potential to influence appetite regulation, energy expenditure, fat metabolism, and overall metabolic efficiency. By engaging multiple hormonal signaling pathways simultaneously, it is being studied for effects that may extend beyond traditional single-receptor GLP-1 agonists.
Researchers are particularly interested in retatrutide’s ability to:
- Support appetite suppression and satiety signaling
- Promote fat loss through enhanced metabolic activity
- Influence energy balance and thermogenesis
- Maintain metabolic flexibility during caloric restriction
Retatrutide represents an advanced area of metabolic research and is commonly evaluated in controlled laboratory environments focused on weight regulation, insulin signaling, and energy homeostasis.
**For research use only. Not for human consumption.
RETA 30MG
CAS: 2381089-83-2
R30 30MG lab-made tri-agonist peptide that works on three hormone receptors: GLP-1, GIP, and glucagon. This triple effect makes it a useful tool for researchers studying metabolism, energy use, and weight control. Scientists use R30 to see how the body manages blood sugar, fat use, and heart function in lab tests.
Researchers receive R30MG as a pure powder, which they mix into a liquid before use. Even though we do not give dosing instructions, scientists can trust the peptide’s high quality, accuracy, and steady results because labs follow strict rules.
Product Care:
Store in a cool, dry place away from light. If Constituted, Please Refrigerate. For longer term storage, freezing at -20°C is recommended to maintain integrity.
Product Note:
All products are shipped in lyphollized or powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.
Product Name: Reta
CAS: 2381089-83-2

Reta 50 MG
GLP-3
Retatrutide s an experimental triple-hormone receptor agonist (GIP, GLP-1, and glucagon) designed for significant weight loss, with Phase 2 trials showing up to 24% reduction in body weight. It is not yet FDA-approved and is only available through clinical trials. Common weekly doses range from 1mg to 12mg.
Before reviewing information about retatrutide, please be aware that this information is for general knowledge only and should not be taken as medical advice. Always consult with a healthcare provider for any health concerns or before making any decisions related to your health or treatment.
Reddit User Experiences With Retatrutide
Many Reddit users report extremely fast weight loss and strong appetite suppression.
Example user comment:
“I started at 350 lbs… now 271 lbs in about 4½ months.”
Another user described dramatic weight loss early in treatment:
“In the first 2 months, I lost 19 kg (about 42 lbs).”
Some people say it significantly improved metabolic health markers during clinical trials:
“BMI went from 34.7 to 23.2… lost 74 lbs.”
Users often mention these benefits:
• strong appetite suppression
• reduced “food noise”
• rapid fat loss
Key Details on Retatrutide:
Weight Loss Efficacy: In studies, participants experienced substantial weight loss, with some studies indicating a 17.5% mean reduction at 24 weeks and up to 24.2% at 48 weeks.
Mechanism: It acts as a triple agonist, targeting GIP, GLP-1, and glucagon receptors to reduce hunger and increase energy expenditure.
Dosage Information: Clinical trial dosages have ranged from 1mg to 12mg. Dosing schedules are typically graduated, starting lower and increasing over several weeks.
Side Effects: The most common side effects are gastrointestinal, including nausea, diarrhea, vomiting, constipation, and abdominal pain.
Safety Considerations: Studies have noted potential, dose-dependent increases in heart rate and mild-to-moderate cardiac arrhythmias.
Important Safety Warning
Retatrutide is currently in clinical trials and has not been approved by the FDA for safety or effectiveness. Products sold online as "retatrutide peptide" are unregulated and not considered safe or legal for consumption.
For more information, you can read the Phase 2 Trial results in The New England Journal of Medicine.
Reta 5MG
Retatrutide (Research Peptide)
Retatrutide is a next-generation investigational peptide studied for its unique triple-agonist activity across the GLP-1, GIP, and glucagon receptors. This multi-pathway mechanism has made retatrutide a subject of growing interest in metabolic and energy-regulation research.
Reddit User Experiences With Retatrutide
Many Reddit users report extremely fast weight loss and strong appetite suppression.
Example user comment:
“I started at 350 lbs… now 271 lbs in about 4½ months.”
Another user described dramatic weight loss early in treatment:
“In the first 2 months, I lost 19 kg (about 42 lbs).”
Some people say it significantly improved metabolic health markers during clinical trials:
“BMI went from 34.7 to 23.2… lost 74 lbs.”
Users often mention these benefits:
• strong appetite suppression
• reduced “food noise”
• rapid fat loss
In preclinical and early clinical research settings, retatrutide has been explored for its potential to influence appetite regulation, energy expenditure, fat metabolism, and overall metabolic efficiency. By engaging multiple hormonal signaling pathways simultaneously, it is being studied for effects that may extend beyond traditional single-receptor GLP-1 agonists.
Researchers are particularly interested in retatrutide’s ability to:
- Support appetite suppression and satiety signaling
- Promote fat loss through enhanced metabolic activity
- Influence energy balance and thermogenesis
- Maintain metabolic flexibility during caloric restriction
Retatrutide represents an advanced area of metabolic research and is commonly evaluated in controlled laboratory environments focused on weight regulation, insulin signaling, and energy homeostasis.
For research use only. Not for human consumption.

Retatrutide 60 MG
Retatrutide (LY3437943) is an investigational once-weekly injectable triple-hormone receptor agonist (GIP, GLP-1, and glucagon) for obesity, type 2 diabetes, and related metabolic conditions. Phase 2 trials showed impressive results, with up to 24.2% weight loss at 48 weeks, making it potentially more effective than current medications like semaglutide.
Reddit User Experiences With Retatrutide
Many Reddit users report extremely fast weight loss and strong appetite suppression.
Example user comment:
“I started at 350 lbs… now 271 lbs in about 4½ months.”
Another user described dramatic weight loss early in treatment:
“In the first 2 months, I lost 19 kg (about 42 lbs).”
Some people say it significantly improved metabolic health markers during clinical trials:
“BMI went from 34.7 to 23.2… lost 74 lbs.”
Users often mention these benefits:
• strong appetite suppression
• reduced “food noise”
• rapid fat loss
• improvements in cholesterol or metabolic markers
One Reddit user summed it up like this:
“Retatrutide really pushed things to the next level.”
Key Aspects of Retatrutide:
Average Weight Loss: Clinical trials (Phase 2) demonstrated that participants on the highest dose (12 mg) achieved an average weight loss of 24.2% (roughly 50+ lbs) over 48 weeks. Recent Phase 3 results showed an average weight loss of up to 71.2 lbs.
Benefits & Mechanism: As a triple-agonist, it acts on GIP, GLP-1, and glucagon receptors to significantly reduce appetite, slow digestion, and increase energy expenditure (fat burning). It is also being studied for knee osteoarthritis, sleep apnea, and fatty liver disease.
Side Effects: Similar to other GLP-1/GIP medications, common side effects include nausea, diarrhea, vomiting, and constipation. Some studies noted transient, mild skin sensations (tingling) and temporary increases in liver enzymes.
Retatrutide is currently in Phase 3 trials and is not yet FDA-approved or commercially available for prescription, though it is considered a major advancement in obesity pharmacotherapy.

Selank 10MG
CAS Number: 129954-34-3
Selank – Research Overview
Chemical name: Thr-Lys-Pro-Arg-Pro-Gly-Pro
Common research name: Selank
Class: Synthetic heptapeptide / Tuftsin analog
Sequence: TKPRPGP
Primary targets: GABAergic modulation (calming neurotransmitter system) and immune-neuropeptide pathways
Molecular weight: ~863 Da
Selank is a synthetic analog of the endogenous peptide tuftsin (a natural immunomodulatory peptide).
It has been investigated in preclinical studies for its effects on:
Anxiety-related behavior (anxiolytic-like effects in animals)
Stress response
Cognitive function (learning and memory in rodent models)
Immune modulation
Monoamine neurotransmission (serotonin, dopamine, norepinephrine systems)
All findings refer to animal and in-vitro models, not established human outcomes.
Molecular & Pharmacological Profile
In experimental systems, Selank has been shown to:
Modulate GABAergic transmission (influences the main calming/inhibitory neurotransmitter)
Affect monoamine levels (serotonin, dopamine, norepinephrine – mood and focus pathways)
Interact with tuftsin receptors (immune and nervous system signaling)
Influence expression of Brain-Derived Neurotrophic Factor (BDNF) in some models (supports neuronal health and plasticity)
Show anxiolytic-like and nootropic-like profiles in rodents (reduced anxiety-like behavior and improved learning in animal tests)
These characteristics make Selank a common subject in neuropsychopharmacology and neuroimmune research.
Mechanism of Action (Preclinical)
Selank’s exact mechanism is still being clarified, but several key preclinical mechanisms have been identified:
- Modulation of GABA System
Selank appears to enhance GABAergic activity
(supporting the brain’s primary calming/inhibitory system),
which may explain its anxiolytic-like (anti-anxiety–like) effects in animal models. - Effects on Monoamine Neurotransmitters
Preclinical data show Selank can:
Influence serotonin metabolism (mood and anxiety regulation)
Affect dopamine and norepinephrine levels (motivation, focus, arousal)
(these are research findings, not clinical claims). - Tuftsin Receptor Interaction
Because it is a tuftsin analog, Selank interacts with tuftsin-sensitive pathways
(bridging immune and nervous system signaling). - Neurotrophic & Gene Expression Effects
Some studies report:
Increased BDNF expression (a key growth factor for neurons)
Changes in expression of genes related to stress response and synaptic plasticity
(how neurons adapt and form new connections) - Immunomodulatory Activity
Selank has been shown to:
Influence cytokine balance (immune signaling molecules)
Shift immune responses toward a more regulated profile in animal models.
Preclinical Research: Potential Applications
Selank has been investigated mainly in rodent and in-vitro models, including:
- Anxiety & Stress-Response Models
Reduced anxiety-like behavior in elevated plus maze and open-field tests
Improved behavioral response under chronic stress conditions
Stabilization of stress-induced changes in neurotransmitters
(These are behavior tests in animals, often used as proxies for anxiety research.) - Cognitive & Nootropic Research
Improved learning and memory performance in maze and conditioning tasks
Enhanced attention and information retention in some paradigms
Support of BDNF and synaptic plasticity–related pathways
(suggestive of pro-cognitive effects in research models) - Neuroimmune & Inflammatory Models
Modulation of pro-inflammatory vs anti-inflammatory cytokines
Potential normalization of immune disturbances under stress
Interaction between nervous and immune systems (neuroimmune axis) - Antidepressant-Like and Anxiolytic-Like Profiles (Preclinical)
Behavioral patterns similar to anxiolytic compounds in animals
Some antidepressant-like effects in rodent tests (forced-swim, tail-suspension–type paradigms)
(Again: preclinical behavioral research, not established human therapy.)
Efficacy Observed in Preclinical Models
Behavioral Studies (Rodents)
Reduced anxiety-like behavior in standard tests (more time in open arms, reduced freezing, etc.)
Enhanced exploratory behavior (animals more willing to explore novel environments)
Better performance on learning and memory tasks (faster acquisition, better recall)
Biochemical / Molecular Findings
Increased GABAergic tone (more inhibitory/calming signaling)
Modulation of serotonin and dopamine turnover
Upregulation of BDNF and other neurotrophic factors in some brain regions
Adjusted cytokine profiles and immune markers under stress
Stress-Model Outcomes
Reduced negative behavioral impact of chronic mild stress
Normalization of certain stress-induced neurotransmitter changes
All of the above are limited to preclinical research.
Safety & Tolerability (Preclinical)
Across available animal research:
Selank has shown low toxicity at standard experimental doses
No major organ toxicity reported in rodent studies
No strong sedative or motor-impairing effects
(animals typically retain normal locomotor activity)
Long-term safety in humans has not been established
Not evaluated or approved by major regulators as a therapeutic agent
Regulatory Status & Disclaimer
Selank is an investigational research peptide.
It is not approved by the FDA or other regulatory agencies for medical, dietary, or cosmetic use.
For Research Use Only.
Not for human consumption.
Not for veterinary use.
Not for diagnostic or therapeutic purposes.
No medical claims are made or implied.
Researchers are responsible for compliance with all applicable laws and regulations in their jurisdiction.
Key References (Preclinical)
Ashmarin I.P. et al. Tuftsin-derived peptides and neuroimmune regulation. Neurosci Behav Physiol.
Neznamov G.G. et al. Selank: Neuropsychopharmacological profile in preclinical models. Bull Exp Biol Med.
Gudasheva T.A. et al. Selank and GABAergic modulation in anxiety models. J Neurochem.
Medvedev A.E. et al. Effects of Selank on BDNF expression and stress response pathways. Neurochem J.
Kozyrev S.A. et al. Immunomodulatory and anxiolytic-like effects of Selank in rodents. Int J Neuropsychopharmacol.
Selank is a synthetic peptide that is known for its anxiolytic (anti-anxiety) and nootropic (cognitive-enhancing) effects. It is a synthetic analog of the naturally occurring peptide tuftsin and was developed in Russia, where it is approved for anxiety and cognitive enhancement, though it is not approved by the U.S. Food and Drug Administration (FDA) for any condition. Potential benefits include reducing anxiety without sedation, improving memory and focus, and supporting mood stability

Semaglutide 10MG
CAS Number: 910463-68-2
Description
GLP S-10MG is a synthetic peptide, is designed to activate GLP-1 receptors, supporting the study of metabolic pathways for research purposes.
Product Care:
Store in a cool, dry place away from light. If Constituted, Please Refrigerate. For longer term storage, freezing at -20°C is recommended to maintain integrity.
Product Note:
All products are shipped in lyphollized or powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.

Semaglutide 20 MG
CAS Number: 910463-68-2
Semaglutide 20mg – Research Grade GLP-1 Peptide
Semaglutide 20mg is a research-grade GLP-1 receptor agonist peptide studied for its role in metabolic regulation, appetite signaling, and body composition pathways. This compound has become one of the most widely researched peptides for its effects on satiety, glucose control, and weight-related outcomes.
???? What is Semaglutide?
Semaglutide is a GLP-1 (glucagon-like peptide-1) analog that mimics a naturally occurring hormone involved in:
Appetite regulation
Insulin signaling
Gastric emptying
Energy balance
Research shows GLP-1 agonists reduce hunger signals and increase feelings of fullness, leading to decreased caloric intake and improved metabolic markers.
⚙️ Key Research Areas
▪️ Appetite & Satiety Signaling
Semaglutide is widely studied for its ability to reduce “food noise” and cravings, helping regulate eating behavior.
▪️ Weight & Body Composition
Clinical research demonstrates average weight reductions of ~10–20% over time when combined with lifestyle factors.
▪️ Glucose & Metabolic Health
Originally developed for metabolic research, semaglutide supports:
Blood sugar regulation
Insulin sensitivity
Reduced metabolic stress
▪️ Extended Systemic Benefits (Emerging Research)
Ongoing studies suggest potential links to:
Cardiovascular health support
Reduced inflammation
Behavioral and reward-system modulation
???? Real-World User Experiences (Reddit Insights)
Here are real, unfiltered user experiences pulled from Reddit discussions:
“I’m 9 weeks in and have lost about 20 lbs… I feel fantastic.”
“I had the typical side effects early… now I have essentially zero side effects.”
“Total loss 116 lbs.”
Users frequently report:
Significant appetite suppression
Steady, sustainable weight loss
Reduced cravings and binge tendencies
However, not all experiences are identical:
⚠️ Observed Research Considerations
Across both clinical and user-reported data:
Common research-reported effects include:
Nausea
Slowed digestion
Fatigue
Outcomes vary based on:
Individual biology
Diet & lifestyle variables
Duration of use
???? Product Details
Compound: Semaglutide
Quantity: 20mg
Form: Lyophilized powder
Purity: Research-grade
Use: Laboratory research only
???? Why Researchers Choose Semaglutide
One of the most studied GLP-1 peptides available
Strong data in metabolic and weight-related pathways
Increasing interest in multi-system effects beyond weight
⚖️ Important Notice
For research use only. Not for human consumption.
This product is intended strictly for laboratory and scientific research purposes.

Semaglutide 5 MG
Product Name: Sema
CAS: 910463-68-2
Molecular Mass: 4114 g/mol
Molecular Formula: C187H291N45O59
PubChem ID: 56843331

Semax 10 MG
Semax 10mg is a synthetic peptide (analogue of ACTH 4-10) commonly supplied as a research compound or for therapeutic use to enhance cognitive function, memory, focus, and neuroprotection by increasing BDNF (brain-derived neurotrophic factor). It acts on neurotransmitter systems, such as dopamine and serotonin, to potentially improve mental clarity, energy, and cognitive resilience against stress.
Reddit User Experiences With Semax
Focus and Cognitive Performance
Some Reddit users report strong improvements in focus and mental clarity.
“Semax was the best. I swear by that stuff… especially focus.”
Another user described it helping with attention and memory:
“It helped immensely… I remember more from the days I take it and focus better.”
Users often describe it as a clean mental stimulation rather than jittery energy.
Mood and Emotional Stability
Some Reddit discussions mention improvements in mood or emotional balance.
Example comment:
“Probably the best thing ever for me personally in cognitive function and mood.”
Another user experimenting with it for emotional symptoms reported:
“It seemed to make me more level headed.”
Brain Fog and Mental Clarity
Semax is often discussed as a brain-fog clearing peptide.
Typical Reddit discussion:
People say it “clears up brain fog and improves focus.”
Some users describe feeling more alert and mentally engaged shortly after taking it.
Anxiety and Depression Improvements
One Reddit user tracked mood scores during a personal experiment:
“Depression score dropped from 12 to 2… anxiety score from 11 to 2.”
Users discussing this peptide frequently say it feels mentally uplifting without being overly stimulating.
Common Positive Themes From Reddit
Across discussions, people who like Semax usually say it helps with:
• mental clarity
• sustained focus
• memory recall
• reduced brain fog
• improved mood
• feeling more “mentally sharp”
Semax is widely described as a nootropic peptide studied for neuroprotective and cognitive effects, particularly in Eastern Europe.
Key Information Potential Effects
Mechanism: Modulates dopaminergic and serotonergic systems, increases BDNF, and boosts acetylcholine.
Benefits: Primarily used for nootropic, neuroprotective, and cognitive-enhancing effects (e.g., focus, memory).
Common Applications: Often used for improving mental clarity, productivity, and recovering from brain circulation issues.
Administration: Typically administered via nasal spray or subcutaneous injection.
Dosage: While research vials are frequently sold in 5 and 10mg quantities, specific dosage protocols should be determined by a healthcare provider.
Side Effects and Considerations
Semax is generally considered to have a low toxicity profile, but potential, usually mild, side effects can include:
Headache
Dizziness
Mild insomnia or anxiety (if overstimulated)
Irritation at the site of administration (nasal spray)
Important Safety Information
Medical Oversight: It is crucial to consult a doctor before starting Semax, especially if you have pre-existing conditions like epilepsy, kidney disease, heart disease, or liver disease.
Not for Everyone: It should not be used by pregnant or breastfeeding individuals without medical advice.
Drug Interactions: It may interact with other medications, particularly those affecting neurotransmitter levels.
Note: The information provided is for educational purposes based on available research and should not be taken as medical advice.

Sermorelin 5 MG
Sermorelin Peptide
Product Overview
Sermorelin is a synthetic peptide analog of growth hormone–releasing hormone (GHRH) that has been widely studied for its ability to stimulate the natural, pulsatile release of endogenous growth hormone (GH). Rather than supplying growth hormone directly, Sermorelin appears to support the body’s own GH signaling pathways by interacting with GHRH receptors in the anterior pituitary.
Because GH release stimulated by Sermorelin remains under physiological feedback control, researchers have shown interest in its potential to promote balanced GH dynamics while avoiding the non-physiological hormone exposure associated with exogenous GH administration.
Research Background & Mechanism
Sermorelin binds to GHRH receptors on pituitary somatotroph cells, triggering intracellular signaling cascades that promote synthesis and secretion of growth hormone. This process appears to preserve the natural circadian rhythm of GH secretion, with peak activity occurring during sleep-associated pulses.
Through downstream GH signaling, Sermorelin has been investigated for its potential involvement in:
- Lean muscle development and maintenance
- Fat metabolism and body composition regulation
- Tissue repair and recovery
- Bone remodeling and structural integrity
- Collagen synthesis and skin support
- Sleep quality and recovery signaling
Unlike some growth hormone secretagogues, Sermorelin does not appear to act via ghrelin receptors and has not been shown to significantly influence appetite or other pituitary hormones.
Potential Research Applications
Based on preclinical and clinical research models, Sermorelin has been explored for its possible role in:
- Supporting age-related decline in GH signaling
- Promoting anabolic balance and nitrogen retention
- Enhancing recovery following physical stress
- Supporting metabolic efficiency and insulin sensitivity
- Investigating GH-related neuroendocrine regulation
Researchers value Sermorelin for its relatively short half-life, which allows for precise timing of GH stimulation without prolonged systemic exposure.
Peptide Specifications
Molecular Formula: C₁₄₉H₂₄₆N₄₀O₄₂
Molecular Weight: ~3357 g/mol
Sequence:
H-Tyr-Ala-Asp-Ala-Ile-Phe-Thr-Gln-Ser-Tyr-Arg-Lys-Val-Leu-Ala-Gln-Leu-Ser-Ala-Arg-Lys-Leu-Leu-Gln-Asp-Ile-Leu-Ser-Arg-Lys-NH₂
Research Considerations
Sermorelin has been studied both independently and alongside other GH-axis peptides for comparative evaluation of pulsatile GH release, tissue response, and metabolic markers. Its mechanism of action makes it a frequent subject of investigation in endocrine and aging-related research models.
Disclaimer
This product is a research chemical intended exclusively for laboratory and in-vitro experimentation. It is not intended for human or animal use. Any form of bodily administration is prohibited. This material is provided for educational and informational purposes only and is available solely to licensed researchers and qualified professionals.

SLUPP-322 5MG
Research:mimic exercise-like metabolic
SLU-PP-332 – Research Overview
Class: Exercise-mimetic small molecule
Primary targets: ERRα (Estrogen-Related Receptor alpha) & ERRγ (Estrogen-Related Receptor gamma)
Category: Metabolic activator / mitochondrial regulator
Research focus: Endurance pathways, mitochondrial biogenesis, oxidative metabolism
SLU-PP-332 is a synthetic small molecule investigated for its ability to activate the cellular programs induced by endurance exercise.
Preclinical studies suggest that SLU-PP-332 influences:
- Mitochondrial biogenesis (more mitochondria)
- Fatty-acid oxidation (burning fat for fuel)
Energy expenditure
- Aerobic/endurance capacity
- Oxidative muscle-fiber pathways
All findings refer to rodent and in-vitro research, not clinical effects.
Mechanism of Action (Preclinical)
SLU-PP-332 functions primarily as a dual ERRα/ERRγ agonist, stimulating two “master switches” that control cellular energy metabolism.
- ERRα Activation (energy-regulation receptor)
- ERRα controls:
- Mitochondrial gene expression
- Fatty-acid transport
- Oxidative phosphorylation (ATP generation)
Activation leads to:
- Increased fat oxidation
- Improved mitochondrial efficiency
- Greater endurance-style metabolic output
-
ERRγ Activation (endurance-receptor pathway)
ERRγ influences:- Slow-twitch (oxidative) muscle programming
- Vascularization
- Long-duration energy systems
Research shows ERRγ activation results in: - Higher aerobic capacity
- Improved fuel efficiency
- Increased mitochondrial content
-
Upregulation of PGC-1α
(The “master regulator” of mitochondrial biogenesis)
SLU-PP-332 boosts expression of PGC-1α, which triggers:- Production of new mitochondria
- Enhanced mitochondrial density
- Increased cellular resilience
- Higher ATP output
-
Fatty-Acid Oxidation Pathway Activation
Rodent studies indicate:- Increased CPT1A and CPT2 activity
(key enzymes for fat-burning) - Higher β-oxidation rates
- Lower reliance on glycolysis (less sugar burning, more fat burning)
- This produces an exercise-like metabolic shift.
- Increased CPT1A and CPT2 activity
-
Exercise-Mimetic Gene Expression
SLU-PP-332 induces a transcriptomic pattern similar to endurance training, including:- Mitochondrial respiratory chain genes
- Oxidative muscle-fiber genes
- Angiogenesis signaling genes
- Stress-adaptation pathways
- Preclinical Research: Potential Applications
SLU-PP-332 has been studied in the following research areas:
-
Endurance & Aerobic Capacity Models
Rodent studies show:- Increased treadmill running distance
- Higher VO₂-related metrics
- Reduced fatigue
- Improved recovery markers
(All in animals, not humans.)
-
Mitochondrial Biogenesis Research
SLU-PP-332 has consistently upregulated:- PGC-1α
- TFAM
- NRF1/NRF2
- These are the genes responsible for generating and repairing mitochondria.
-
Metabolic Flexibility Studies
In studies focused on metabolic switching:- Animals burned more fat at rest
- Respiratory quotient (RQ) shifted toward fatty-acid usage
- Lower lactate production under physical stress
(Suggests a more efficient “endurance metabolism.”)
-
Muscle-Fiber Research
Preclinical models found:- Increased oxidative slow-twitch fiber markers
- Higher mitochondrial density within muscle cells
- Improved contractile endurance
-
Lipid & Glucose Metabolism
SLU-PP-332 has shown:- Increased fat mobilization
- Improved lipid oxidation
- Lower lipid accumulation in metabolic-stress models
- More stable blood glucose responses under activity
(Mechanism related to ERR pathways, not therapeutic claims.)
Efficacy Observed in Preclinical Studies
Endurance Findings
- 20–50% increased running time in rodents
- Greater maximum distance
- Higher fatigue resistance
- Improved aerobic efficiency
Mitochondrial Effects
- Strong upregulation of mitochondrial genes
- Increased mitochondrial respiration
- Higher ATP production
- Improved mitochondrial membrane potential
Metabolic Effects
- Shift toward fat-as-primary-fuel
- Increased oxygen consumption efficiency
- Better metabolic flexibility
Safety & Tolerability (Preclinical)
- Well tolerated in rodent models
- No major organ toxicity reported at research doses
- Non-hormonal mechanism
- Effects mediated through metabolic receptors
- Long-term human safety unknown
- Not FDA-approved
Regulatory Status & Disclaimer
- SLU-PP-332 is an investigational research compound.
- It is not approved by the FDA or any regulatory body for therapeutic, dietary, or medical use.
- For Research Use Only.
- Not for human consumption.
- Not for veterinary use.
- Not for diagnostic or therapeutic applications.
- No medical claims are made or implied.
Key References (Preclinical)
Ma T. et al. ERR agonists as exercise mimetics in metabolic research. Cell Reports.
Schreiber S. et al. Regulation of mitochondrial biogenesis by ERR signaling. PNAS.
Narkar V. Exercise-mimetic molecular pathways. Cell.
Nedungadi P. et al. ERRγ activation and endurance metabolism. Nat Metabolism.
Supplemental ERR agonist data across metabolic rodent models.

SLUPP-332 10MG
CAS NO. 303760-60-3
SLU-PP 332 10MG a synthetic research compound (an "exercise mimetic") that acts as a pan-agonist of the estrogen-related receptors (ERRα, ERRβ, and ERRγ). It is being researched for its potential therapeutic use in treating metabolic diseases such as obesity, type 2 diabetes, and heart failure by mimicking the metabolic effects of exercise.
Reconstitution Note: Many Users First Dissolve It in DMSO A typical Reddit method described:
“Dissolve 5 mg in about 0.5 ml DMSO… then slowly add bac water.” r/BodyHackGuide
SLU-PP-332 – Research Overview
Chemical class: Exercise-mimetic small molecule
Primary target: ERRα / ERRγ (Estrogen-Related Receptor alpha & gamma)
Category: Metabolic activator / Mitochondrial regulator
Research focus: Muscle metabolism, endurance pathways, mitochondrial biogenesis
Molecular type: Synthetic non-peptide small molecule
SLU-PP-332 is an ERRα/ERRγ agonist, investigated in preclinical studies for its ability to mimic the cellular effects of exercise by activating energy-regulation pathways.
It has been examined in rodent and cellular research for effects on:
- Endurance capacity
- Mitochondrial biogenesis (creation of new mitochondria)
- Fatty-acid oxidation (fat burning)
- VO₂-related metabolic pathways
- Muscle-fiber energy use
All findings are from preclinical models, not confirmed human outcomes.
Molecular & Pharmacological Profile
SLU-PP-332 belongs to a class of compounds called exercise mimetics — molecules that activate the same cellular programs triggered by physical activity.
In research settings, SLU-PP-332 has been shown to:
- Activate ERRα and ERRγ receptors(master regulators of mitochondrial energy metabolism)
- Increase PGC-1α signaling(the key coactivator for endurance + mitochondrial growth)
- Enhance fatty-acid oxidation(increased fat → energy conversion)
- Shift skeletal muscle toward oxidative metabolism (endurance-style muscle activity)
- These changes resemble the molecular signature of aerobic exercise.
Mechanism of Action (Preclinical)
- ERRα / ERRγ Activation (exercise-related transcription factors)
These receptors control:- Mitochondrial activity
- Glucose & fat utilization
- Oxidative energy production
- SLU-PP-332 acts as an agonist, turning these pathways “on.”
- Increased PGC-1α Expression
(PGC-1α = “mitochondrial master switch”)
This leads to:- More mitochondria
- More efficient mitochondria
- Greater endurance-style cellular activity
- Enhanced Oxidative Metabolism
Preclinical studies show:- Higher reliance on fatty-acid oxidation
- Improved metabolic efficiency
- Increased ATP production(better sustained cellular energy)
- Exercise-Mimetic Gene Expression
- SLU-PP-332 induces gene patterns similar to those activated by:
- Endurance training
- Aerobic conditioning
- Mitochondrial stress adaptation
- Improved Fat Utilization
Rodents given SLU-PP-332 showed increased use of fat as the primary energy source.
Preclinical Research: Potential Applications
SLU-PP-332 is studied for its effects on muscle metabolism and endurance. - Endurance & Performance Models
- In rodent experiments, SLU-PP-332:
- Increased treadmill running time
- Increased distance covered
- Enhanced VO₂ max–related markers(oxygen-utilization pathways in muscle)
- These findings reflect exercise-mimetic effects at the cellular level.
- Mitochondrial Biogenesis Research
SLU-PP-332 upregulated:- PGC-1α
- TFAM
- NRF1/NRF2(all major mitochondrial-production genes)
This led to: - More mitochondria
- Higher mitochondrial capacity
- Improved oxidative phosphorylation (ATP production)
- Metabolic Health & Energy-Utilization Models
- Preclinical data show:
- Reduced reliance on glucose
- Increased fatty-acid utilization
- Greater metabolic flexibility (switching fuels more efficiently)
- Improved muscle-fiber oxidative phenotype
(shift toward slow-twitch/endurance muscle characteristics)
- Exercise-Mimetic Signaling
The most notable finding across multiple studies:- SLU-PP-332 activates molecular pathways nearly identical to those activated by endurance exercise — even in sedentary animals.
This includes: - AMPK-adjacent signaling
- Enhanced mitochondrial gene transcription
- Increased oxidative gene expression
- SLU-PP-332 activates molecular pathways nearly identical to those activated by endurance exercise — even in sedentary animals.
Efficacy Observed in Preclinical Models
Rodent endurance studies:
- Increased treadmill endurance by 20–50% (varied by study)
- Higher resistance to fatigue
- Better overall aerobic performance
Mitochondrial markers:
- Increased mitochondrial density in muscle tissue
- Higher ATP production capacity
- Upregulated oxidative phosphorylation complexes
Metabolic effects:
- Increased fat oxidation
- Reduced lactate accumulation
- Lower respiratory quotient (suggesting fat-dominant metabolism)
Muscle physiology:
- Transition toward oxidative muscle fibers (slow-twitch phenotype)
- Improved mitochondrial membrane potential
- Enhanced mitochondrial enzyme activity
All findings in animals, not humans.
Safety & Tolerability (Preclinical)
- Generally well-tolerated at experimental doses
- No major toxicity reported in short-term rodent studies
- No evidence of hormonal disruption
- Mechanism is metabolic, not endocrine
- Long-term safety, human pharmacokinetics, and clinical tolerability remain unknown
- SLU-PP-332 is not approved for clinical use.
Regulatory Status & Disclaimer
- SLU-PP-332 is an investigational research chemical.
- It is not approved by the FDA or any regulatory agency for therapeutic, dietary, cosmetic, or medical use.
For Research Use Only.
Not for human consumption.
Not for veterinary use.
Not for diagnostic or therapeutic purposes.
No medical claims are made or implied.
Researchers must comply with all applicable regulations.
Key References (Preclinical)
Ma T. et al. Exercise-mimetic effects of ERR agonist SLU-PP-332 in rodent models. Cell Reports.
Dittenhafer-Reed K.E. ERR-mediated mitochondrial regulation in metabolic tissues. Nat Metab.
Schreiber S.N. et al. ERRγ and oxidative metabolism regulation. PNAS.
Narkar V.A. et al. Molecular signatures of exercise mimetics. Cell.
Supplementary mitochondrial gene-expression data from ERR agonist studies.
Each vial of SLU-PP-332 comes as a freeze-dried powder to keep its quality and shelf life. Before testing, researchers mix the powder with a sterile solution. Every batch meets strict lab standards for purity and consistency, so results stay clear and reliable.
All products are shipped in lyophilized or powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.
SS-31
Mitochondrial Research Peptide
SS-31 (also known as Elamipretide, Bendavia, or MTP-131) is a research peptide that selectively targets mitochondria. It is an investigational drug not approved by the FDA for general human therapeutic use, and information on its use is for research or educational purposes only.
Research Considerations
Information regarding the use of SS-31 stems primarily from preclinical studies and clinical trials, where parameters such as administration method (intravenous or subcutaneous) and the condition being studied influenced research protocols. Data on long-term safety in humans beyond a few weeks of treatment remains limited.
Potential Benefits Explored in Research
SS-31 functions by accumulating in the inner mitochondrial membrane and interacting with cardiolipin. This interaction is thought to help stabilize mitochondrial structure, improve the efficiency of the electron transport chain, decrease the production of reactive oxygen species, and enhance the synthesis of ATP, a key energy molecule.
While numerous preclinical studies, primarily in animal models, have explored a wide range of potential benefits, outcomes from clinical trials in humans for conditions such as heart failure and mitochondrial myopathy have been varied or inconclusive.
Cardiovascular System: Research in animal models suggests potential protective effects against injury from lack of blood flow, improved exercise capacity, enhanced heart function in models of heart failure, and a reduction in the development of fatty deposits in arteries.
Neurological Function: Studies in animal models indicate possible benefits in reducing age-related cognitive decline, offering protection against damage from traumatic brain injury and stroke, and potentially addressing aspects of Alzheimer's and Parkinson's diseases.
Musculoskeletal System: Preclinical research has suggested that SS-31 may reverse declines in muscle function and endurance associated with aging, potentially making muscles more resistant to fatigue.
Renal System: Research in animal models shows potential protective effects against various forms of kidney injury, such as diabetic kidney disease and damage from lack of blood flow followed by reperfusion, by reducing cellular stress and inflammation.
Cellular Aging: By targeting the decline of mitochondrial function often associated with aging, SS-31 is being investigated for potential effects on cellular aging and extending the period of good health at a cellular level.
Note: The details presented here summarize findings from research. SS-31 is classified as a research chemical, and its use should only occur under the guidance of qualified medical professionals or within the framework of formal research.
Disclaimer: This information is intended for general knowledge and should not be interpreted as medical advice. Always consult with a healthcare provider before considering any new treatment or supplement.
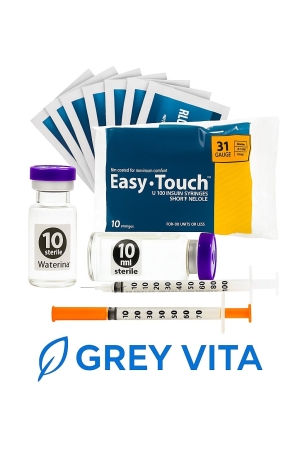
Starter Kit
10ML BacWater, 10 Alcohol Wipes, 10 pk Syringes
Starter Pack for Research.
- 10 Pk 5/16 31 G .5ml Insulin Needles
- 10 alcohol wipes
- 10 ml vial of bac water
Starter Kit (small)
2 Syringes, 2ml sterile water, 2 alcohol wipes.
Small Starter Pack - 2 ml Sterile Water, 2 .5ml Syringes, 2 Alcohol Wipes

T15 - TIRZEPATIDE 15 MG
CAS number for Tirz is 2023788-19-2.
All products are shipped in lyphollized or powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.
Tirzepatide – Research Overview
Class: Dual incretin receptor agonist
Targets:
GIP receptor (GIPR) – glucose-dependent insulinotropic polypeptide
GLP-1 receptor (GLP-1R) – glucagon-like peptide-1
Structure: Synthetic peptide with fatty-acid side chain for extended half-life
Research focus: Metabolic regulation, incretin signaling, insulin pathways, glucagon suppression
Tirzepatide is a dual incretin mimetic designed to activate both GIP and GLP-1 receptors.
It has been studied in rodent and non-human primate research models for its effects on:
- Glucose control
- Energy balance
- Appetite-regulation pathways
- Body-weight regulation
- Incretin hormone signaling
- Insulin secretion and glucagon suppression
All findings refer strictly to preclinical data, not established human outcomes.
Mechanism of Action (Preclinical)
Tirzepatide works by stimulating two incretin receptors simultaneously, creating a multi-layered metabolic signal.
- GIPR Activation (Primary Pathway)
GIP plays major roles in:- Insulin secretion (increased insulin when glucose is high)
- Lipid metabolism
- Adipocyte signaling (fat-cell hormone communication)
Preclinical effects:
- Enhanced glucose-dependent insulin release
- Improved insulin sensitivity
- Reduced adipose inflammation (less inflammatory signaling in fat tissue)
- GLP-1R Activation (Secondary Pathway)
- GLP-1 regulates:
- Insulin secretion
- Glucagon suppression
- Gastric emptying
- Appetite signaling
Preclinical effects:
- Reduced glucagon output
- Slower gastric emptying (food leaves stomach more slowly)
- Lower appetite-signaling activity in the hypothalamus
- Improved glucose tolerance
- Dual-Incretin Synergy
- The key feature of tirzepatide is combined activation of GIPR + GLP-1R.
- In preclinical metabolic studies, dual action has shown:
- Greater metabolic effect than either pathway alone
- More robust insulinotropic response
- Improved energy expenditure markers
- Synergistic appetite-regulation signals
(The “twin incretin” activity is stronger than GLP-1 alone in most rodent models.)
Metabolic Effects Observed in Preclinical Models
-
Glucose Homeostasis
Rodent studies show:- Increased glucose-dependent insulin secretion
- Reduced fasting and post-meal glucose excursions
- Improved β-cell stress markers (better pancreatic function)
- Decreased glucagon levels during hyperglycemia
- Enhanced whole-body insulin sensitivity
-
Body-Weight Regulation Pathways
- Preclinical findings include:
- Reduced food intake
- Lower activation of hypothalamic hunger pathways (ghrelin + AGRP neuron reduction)
- Increased energy expenditure in brown adipose tissue
- Higher fatty-acid oxidation (fat burning)
- Reduced lipogenesis (new fat stored)
-
Adipose Tissue Biology
In rodent adipocyte studies:- Lower inflammation in fat depots (reduced IL-6, TNF-α)
- Improved adiponectin signaling (fat-cell hormone that improves metabolism)
- Reduced hypertrophic adipocytes (smaller fat cells)
-
Liver & Lipid Findings
- Tirzepatide has been evaluated in models of:
- Hepatic fat accumulation
- Lipotoxicity
- Nonalcoholic fatty liver markers
Preclinical results show:
- Reduced triglyceride accumulation
- Improved hepatic insulin signaling
- Better mitochondrial β-oxidation of fats
- Efficacy Observed in Preclinical Studies
- Metabolic Profile
- Strong insulinotropic response when glucose is elevated
- Greater suppression of glucagon compared to single-pathway incretins
- Improved glucose disposal rates
- Lower inflammatory cytokine markers
Appetite & Weight
- Reduced caloric intake
- Lower preference for high-fat diets in rodent choice studies
- Increased satiety signaling
- Significant weight-loss effects in diet-induced obese animals
Cardiometabolic Markers
- Lowered triglycerides and cholesterol in rodent models
- Improved endothelial NO signaling (supports vascular tone)
Reduced oxidative stress markers
(All results from animal/cell studies, not human therapy.)
Safety & Tolerability (Preclinical)
- Generally well-tolerated in animal models
- GI slowing observed due to GLP-1R activity
- No major organ toxicity at research doses
- Long-term safety and PK/PD in humans remain unestablished
- Not FDA-approved for research outside regulated clinical programs
Regulatory Status & Disclaimer
- Tirzepatide is an investigational research peptide.
- It is not approved by the FDA or any regulatory agency for therapeutic, dietary, or cosmetic use.
- For Research Use Only.
- Not for human consumption.
- Not for veterinary use.
- Not for diagnostic or therapeutic applications.
- No medical claims are made or implied.
- Researchers are responsible for appropriate use under local regulation.
Key References (Preclinical)
Wilson J. Dual GIP/GLP-1 receptor activation in metabolic regulation. Cell Metabolism.
Coskun T. Mechanistic profile of tirzepatide in rodent models. Diabetes.
Samms R. GIP receptor biology and energy balance. Endocrinology.
Pfluger P. Incretin hormone synergy and metabolic control. Nat Rev Endocrinol.
Gribble F. Gut peptide signaling in metabolism. J Physiol.
Research Use Only
This product is intended strictly for laboratory and in vitro research applications. T15 is NOT for human or veterinary use, diagnostic purposes, or therapeutic applications. Must be handled by qualified professionals in a controlled setting.
Product Care:
Store in a cool, dry place away from light. If reconstituted, Please Refrigerate. For longer term storage, freezing at -20°C is recommended to maintain integrity.
Product Note:
All products are shipped in lyophilized or original powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.

TB500/BPC 5MG/5MG
BPC-157 + TB-500 – Research Overview
Class: Regenerative-support peptide combination
Components:
BPC-157 – gastric-derived cytoprotective peptide
TB-500 (Thymosin Beta-4 fragment) – actin-binding + cell-migration peptide
Primary research focus:
- Tissue-repair models, angiogenesis (blood-vessel formation), cellular migration, inflammation modulation, and wound-healing pathways in preclinical systems.
- This combination brings together two peptides often studied for complementary roles in regeneration, structural repair, and inflammatory responses in rodent and in-vitro research models.
Why BPC-157 + TB-500 Are Studied Together
- Each peptide affects different parts of the tissue-repair process:
BPC-157 - Enhances angiogenesis (formation of new blood vessels)
- Supports fibroblast activity (connective-tissue building cells)
- Modulates inflammation
- Stabilizes cellular junctions (keeps cells connected tightly)
- Shows cytoprotective effects in GI and vascular models
TB-500
- Promotes cell migration (movement of repair cells to injury sites)
- Increases actin polymerization (supports cell structure/mobility)
- Enhances angiogenesis
- Supports wound repair in muscle/tendon models
- Encourages anti-inflammatory signaling
Together, the combination is researched for producing a broader and more robust set of repair-related signals in animal models.
Mechanisms of Action (Preclinical)
-
Amplified Angiogenesis
Both peptides activate pathways that increase vascular growth (more blood supply → more oxygen/nutrients to the affected area). -
Enhanced Cell Migration & Growth
TB-500 increases actin dynamics → cells move faster
BPC-157 increases fibroblast recruitment → more repair activity -
Coordinated Inflammatory Modulation
Research models show:
-Lower pro-inflammatory cytokines
-Higher repair-associated cytokines(more balanced inflammation response) -
Cytoprotective Effects
- BPC-157 protects vascular and mucosal tissue in preclinical studies, reducing cellular damage markers.
-
Accelerated Tissue Organization
-Both peptides support structural repair:
-Collagen alignment
-Matrix reorganization
-Improved tissue architecture in rodent models
(Better organization means stronger, more functional tissue when examined microscopically.)
Preclinical Research: Potential Applications
-
Musculoskeletal Research
Rodent studies show effects on:
Tendon and ligament repair markers
Muscle-fiber regeneration
Collagen deposition and alignment
Reduced inflammatory lesions in connective tissue -
Soft-Tissue & Wound-Healing Models
Combination research demonstrates:
Faster epithelial closure (wounds closing more quickly)
Better tissue granulation
Increased angiogenesis -
Vascular Models
Improved blood-vessel stability
Enhanced endothelial repair
(endothelial = blood-vessel lining)
Normalization of NO (nitric oxide) signaling pathways -
Inflammatory & Immune Modulation
Preclinical models show:
Reduced cytokine levels (less inflammatory markers)
Modulated T-cell and macrophage responses
Improved immune balance under stress -
Organ-Protection Models
Particularly with BPC-157:- Reduced oxidative stress markers
- Improved structural preservation in injured tissues
- Cytoprotection of GI, liver, and vascular tissue in rodent injury models
TB-500 contributes through its cell migration and angiogenesis roles.
- Efficacy Observed in Preclinical Models
Muscle & Tendon Research
- Increased fibroblast density (more repair cells)
- Stronger collagen fiber arrangement
- Improved biomechanical properties in animal tendon models
Wound & Tissue Healing
- Accelerated wound closure in rodents
- Higher angiogenic markers (VEGF, FGF pathways)
- Richer granulation tissue formation
- Anti-Inflammatory Effects
- Downregulation of TNF-α, IL-6
- Reduced tissue necrosis
- Less inflammatory cell infiltration
Vascular & Endothelial Findings
- Stabilization of tight junctions (BPC-157)
- Improved endothelial migration (TB-500)
- Increased microcirculation
All results pertain only to preclinical work.
Safety & Tolerability (Preclinical)
Both peptides show low toxicity in rodent studies
No major adverse events at typical research doses
No significant organ toxicity reported
TB-500 has shown good tolerability even in long-term exposure models
Long-term human safety is not established
The blend is not FDA-approved.
Regulatory Status & Research Disclaimer
BPC-157 and TB-500 are investigational research peptides.
They are not approved by the FDA or any regulatory agency for therapeutic, dietary, or cosmetic use.
For Research Use Only.
Not for human consumption.
Not for veterinary use.
Not for diagnostic or therapeutic purposes.
No medical claims are made or implied.
Researchers are responsible for compliance with applicable laws.
Key References (Preclinical)
Sikiric P. et al. BPC-157 and angiogenesis in rodent tendon models. Curr Pharm Des. Vukojević J. et al. Cytoprotective roles of BPC-157 in vascular models. J Physiol.
Goldstein A.L. Thymosin beta-4 and tissue regeneration pathways. Ann N Y Acad Sci.
Malinda K. et al. TB-500 (TB4 fragment) and angiogenesis modulation. J Cell Sci.
Seiwerth S. et al. Tissue-repair mechanisms activated by BPC-157. Exp Bio Med.

TESAMORELIN 10MG
Primary CAS number: 218949-48-5
Tesamorelin is a synthetic growth hormone-releasing hormone (GHRH) peptide that contains the entire GHRH amino acid sequence and a trans-3 hexenoic group to protect it from enzymatic degradation, which makes it more stable and extends its half-life.
Tesamorelin – Research Overview
Class: Growth Hormone–Releasing Hormone (GHRH) analog
Sequence: Modified 44–amino acid peptide
Primary target: GHRH receptor (on pituitary somatotrophs)
Research focus: GH secretion, IGF-1 pathways, metabolic signaling, lipid mobilization
Tesamorelin is a stabilized analog of GHRH designed to resist degradation and provide stronger, longer-lasting stimulation of natural GH pulsatility in preclinical models.
It has been investigated for its effects on:
- Growth hormone secretion
- IGF-1 production
- Lipid mobilization
- Visceral adipose signaling
- Metabolic pathways
- Endocrine axis dynamics (how hormone feedback loops adjust and communicate)
All findings refer to animal and cellular research, not established human outcomes.
Mechanism of Action (Preclinical)
Tesamorelin activates the GHRH receptor, resulting in enhanced GH release through natural physiological pathways.
-
GHRH Receptor Activation
Tesamorelin binds to pituitary GHRH receptors, leading to:- Increased amplitude of GH pulses
- Increased frequency of GH pulses
- Enhanced downstream IGF-1 output
- Preservation of normal GH feedback loops
(Stimulation follows the body’s natural rhythm rather than constant GH elevation.)
-
Amplified GH → IGF-1 Axis
Growth hormone triggers the liver and tissues to produce IGF-1, which participates in:- lipid metabolism
- protein synthesis
- cellular turnover
- tissue repair pathways
- Tesamorelin boosts IGF-1 levels more significantly than shorter GHRH fragments (like CJC-1295 without DAC).
-
Visceral Adipose Research Effects
Preclinical models show Tesamorelin influences:- lipolysis (breaking down stored fat)
- reduced triglyceride storage
- improved mitochondrial fat-metabolism gene expression
- GH-mediated lipid oxidation
-
Metabolic Signaling
GH and IGF-1 affect multiple metabolic systems:- Increased fatty-acid oxidation (fat → energy)
- Improved glucose uptake in muscle
- Modified insulin-signaling pathways
- Enhanced mitochondrial function
These remain preclinical findings.
Preclinical Research: Potential Applications
Tesamorelin is commonly studied for its impact on metabolic, endocrine, and tissue-repair systems.
-
Lipid & Adipose Tissue Research
- Research shows Tesamorelin may:
- Reduce visceral adipose markers
- Increase lipolytic enzyme activity
- Improve fatty-acid mobilization
- Normalize adipocyte (fat-cell) signaling
-
Endocrine & GH/IGF Studies
Tesamorelin is a strong model compound for studying:- GH pulsatility
- Pituitary responsiveness
- GHRH receptor pharmacodynamics
- IGF-1–mediated tissue pathways
- It produces some of the highest GH pulses among GHRH analogs in rodent models.
-
Muscle & Tissue Remodeling
GH/IGF-1 have been associated with:- increased collagen turnover
- improved muscle-protein synthesis
- enhanced recovery markers in rodents
- Tesamorelin provides a potent model for studying these pathways.
-
Cognitive & Neurological Pathways
GH and IGF-1 influence:- neurogenesis
- synaptic plasticity
- BDNF signaling
- cognitive stress adaptation
Tesamorelin’s upregulation of GH/IGF-1 makes it a useful tool for exploring these systems in animals.
-
Cardiometabolic Research
In rodent models tesamorelin has been linked to:- reduced triglycerides
- improved lipid oxidation
- decreased inflammatory cytokines (e.g., TNF-α, IL-6)
- improved mitochondrial function
Again — all preclinical, not clinical outcomes.
Efficacy Observed in Preclinical Studies
- Growth Hormone Signaling
- Strong GH pulse generation
- Large increases in IGF-1
- Sustained pituitary activation
Metabolic Outcomes - Decreased visceral-fat markers
- Increased fatty-acid oxidation
- Reduced inflammatory cytokines
- Improved mitochondrial respiratory capacity
- Tissue Remodeling
- Enhanced collagen synthesis
- Upregulated repair genes
- Increased myofibril turnover (basically the cleaning and rebuilding cycle of muscle tissue.)
Lipid Profile - Reduced triglyceride accumulation
- Improved adipocyte insulin signaling
- Increased expression of fat-oxidation enzymes
Safety & Tolerability (Preclinical)
- Well tolerated in rodent models
- No major organ toxicity at standard experimental doses
- Expected GH-related metabolic shifts
- No direct receptor activation outside GHRH pathways
- Long-term human safety unknown
- Tesamorelin is not FDA-approved outside regulated clinical contexts.
Regulatory Status & Disclaimer
- Tesamorelin is an investigational research peptide.
- It is not approved by the FDA or any regulatory agency for therapeutic, dietary, or cosmetic use.
- For Research Use Only.
- Not for human consumption.
- Not for veterinary use.
- Not for diagnostic or therapeutic purposes.
- No medical claims are made or implied.
Key References (Preclinical)
Falutz J. GHRH analogs in GH pulsatility and metabolic pathways. Endocrinology.
Stanley T. Tesamorelin and visceral adipose regulation. Metabolism.
Ionescu M. Pituitary responsiveness to GHRH analogs. Neuroendocrinology.
Gelato M. GH/IGF-1 axis activation via stabilized GHRH analogs. J Clin Invest.
Nogueira J. Tesamorelin and lipid-metabolism models. J Lipid Res
Product Care:
Store in a cool, dry place away from light. If Constituted, Please Refrigerate. For longer term storage, freezing at -20°C is recommended to maintain integrity.
Product Note:
All products are shipped in lyphollized or powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.
Product Name: Tesa***
CAS: 901758-09-6
Molecular Mass: 5136 g/mol
Molecular Formula: C221H366N72O67S
PubChem ID: 483927426
Storage: Peptides should be stored in a dry, cool, dark place. For best preservation, store at 4°C or colder away from bright light.
IPAMORELIN VS TESAMORELIN COMPARISON
Tesamorelin and Ipamorelin are both peptide-based agents in the broader family of growth hormone–releasing peptides (GHRPs). They act through different receptors and are used for different primary purposes, but each ultimately raises natural growth hormone (GH) levels, so they share many downstream effects. Interest in both has grown alongside the surge of peptide-driven weight-loss research sparked by compounds such as semaglutide and liraglutide. Although Tesamorelin and Ipamorelin are often discussed in the context of body composition, their potential reaches well beyond changes in fat mass and lean tissue.
Tesamorelin vs Ipamorelin: Properties
Tesamorelin
Tesamorelin is FDA-approved for HIV-associated lipodystrophy, a very specific indication that does not fully reflect its broader impact on body composition. Studies suggest Tesamorelin can decrease fat mass—especially visceral fat—support muscle and bone mass, enhance peripheral nerve repair, and may even have benefits in mild cognitive impairment.[1]
Ipamorelin
Ipamorelin advanced to phase II clinical trials as a candidate treatment for postoperative ileus. It is one of the most selective agonists of the ghrelin (growth hormone secretagogue) receptor identified to date. In animals, it substantially elevates plasma GH without altering levels of prolactin, follicle-stimulating hormone, luteinizing hormone, thyroid-stimulating hormone, or adrenocorticotropic hormone.[2],[3] This exceptional specificity makes Ipamorelin a powerful tool for studying the effects of increased GH with minimal interference from other hormones.
Tesamorelin vs Ipamorelin: Mechanism of Action
One of the most interesting aspects of these peptides is that they raise GH through different receptor systems.
Ipamorelin
Ipamorelin binds to and activates ghrelin receptors in the brain and pituitary. This triggers GH release via the growth hormone secretagogue receptor (GHSR). Unlike some other GHRPs, it does not act directly on the GHRH receptor.
Tesamorelin
Tesamorelin is a synthetic analog of human growth hormone–releasing hormone (GHRH). By binding to the GHRH receptor on pituitary cells, it initiates intracellular signaling that increases GH synthesis and secretion. Importantly, Tesamorelin promotes GH release in a pulsatile fashion, closely mirroring the body’s natural rhythm of GH secretion.
Both peptides ultimately increase IGF-1, as GH stimulates the liver and other tissues to produce insulin-like growth factor-1. Ipamorelin produces short-lived but pronounced rises in GH and IGF-1, whereas Tesamorelin, typically dosed once daily, causes robust yet transient increases in these hormones. In both cases, GH and IGF-1 levels return to baseline within a few hours, and each preserves the physiological pulsatile pattern of GH release, which is thought to reduce desensitization and side-effects relative to continuous GH stimulation.
Tesamorelin vs Ipamorelin: Fat Loss
The most obvious comparison between Tesamorelin and Ipamorelin is their impact on fat loss and body composition. Tesamorelin is specifically approved for reducing excess fat, but since both peptides stimulate GH, similar changes in body composition are expected.
Tesamorelin
Clinical research shows that Tesamorelin can reduce adiposity by roughly 20% over one year, with particularly strong effects on visceral fat.[1] Visceral adipose tissue, which surrounds the abdominal organs, is more strongly associated with cardiometabolic risk than subcutaneous fat. Excess visceral fat contributes to elevated triglycerides and cholesterol. Tesamorelin administration has been shown to lower triglycerides by about 25% over 26 weeks—results comparable to some standard lipid-lowering medications such as niacin and certain statins.
Although Tesamorelin’s primary indication centers on reducing visceral adipose tissue in HIV-associated lipodystrophy, clinical trials also report improvements in overall body composition, including increases in lean mass. Resistance training combined with Tesamorelin may further enhance these anabolic, muscle-preserving effects.
Ipamorelin
In mice, Ipamorelin can raise GH levels between three- and thirteen-fold above baseline.[2],[3] Animal studies examining body composition indicate that, with diet held constant, Ipamorelin treatment can increase muscle mass by around 9% and decrease fat mass by about 14%, even without changes in exercise volume.
Human data on Ipamorelin and triglycerides are limited. However, by boosting GH, Ipamorelin is expected to influence lipid metabolism through increased lipolysis and potential improvements in lipid profiles. In animal models, reductions in fat mass from Ipamorelin could secondarily improve triglyceride levels.
Tesamorelin vs Ipamorelin: Fat Quality
Beyond the amount of fat, researchers now emphasize fat quality—how adipose tissue functions at a cellular and metabolic level. Adipose tissue (AT) is not just storage; it is an active endocrine and inflammatory organ composed of adipocytes, connective tissue, nerves, vasculature, and immune cells. It secretes hormones and cytokines such as leptin and adiponectin that influence systemic metabolism and cardiovascular risk.
VAT (visceral adipose tissue) and SAT (subcutaneous adipose tissue) differ in their metabolic impact. Moreover, the same quantity of fat can vary in quality. CT imaging can assess fat density: more dense fat usually reflects smaller, healthier adipocytes, whereas lower density suggests enlarged, lipid-packed cells that are more inflamed and metabolically harmful.[4]
Population studies like the Framingham Heart Study show that lower VAT and SAT density correlates with reduced adiponectin and higher cardiovascular risk.[4] In simple terms, low-quality fat is made up of hypertrophic adipocytes that are inflamed and metabolically inflexible.
Tesamorelin
Tesamorelin appears to improve not only fat quantity but also fat quality. In subjects receiving Tesamorelin, triglycerides and other lipid parameters improved, adipocytes became smaller and denser, and adiponectin levels rose.[5] This indicates that Tesamorelin reduces fat mass while also making remaining adipose tissue healthier and more metabolically favorable.
Comparable trials have not yet been done for Ipamorelin, but given that both molecules are GH secretagogues, it is plausible that Ipamorelin could exert similar effects on adipose tissue quality. More research is needed to confirm this and to better define how GH secretagogues may influence diabetes, cardiovascular disease, and other conditions linked to dysfunctional adipose tissue.
Importantly, discussion of fat quality does not imply that carrying excess fat is healthy. Rather, it underscores that all bodies—lean or overweight—contain adipose tissue, and that the health of that tissue matters. Future work on fat quality may yield strategies to improve metabolic health and longevity even in individuals who are already relatively lean.[4],[5]
Tesamorelin vs Ipamorelin: Muscle Density
GH secretagogues are known to increase muscle and bone mass and decrease fat mass, but they also influence the quality of these tissues.
Ipamorelin and Bone
Ipamorelin has been extensively studied for its effects on bone. Clinical investigations have evaluated it for conditions like osteoporosis and corticosteroid-induced bone loss. Traditional treatments (e.g., bisphosphonates) help prevent further bone loss and increase bone mineral density, but they do not necessarily restore healthy bone architecture. Ipamorelin, by contrast, stimulates the growth and proliferation of bone-forming cells, boosting bone formation by up to 400% in some settings. This leads to improvements in both bone density and bone quality and may avoid complications associated with some current therapies. Ipamorelin offers these benefits while being less costly than many standard treatments.
Tesamorelin and Muscle
Given Ipamorelin’s effect on bone quality, researchers asked whether other GH secretagogues could similarly improve the quality of other tissues. Tesamorelin, already approved for human use, became a logical candidate. Studies show Tesamorelin reduces intramuscular fat and increases muscle cross-sectional area, effectively improving muscle density.[6],[7] Intramuscular fat accumulation is linked to impaired physical function, frailty, and even a higher risk of falls, particularly in older adults.
Tesamorelin’s ability to shrink intramuscular fat depots and expand muscle area suggests it promotes myofibrillar hypertrophy—growth of the actual muscle fibers. Higher muscle density correlates with greater strength, better endurance, and resilience to aging-related decline.[6],[7]
Tesamorelin vs Ipamorelin: Pain and Inflammation
Ghrelin mimetics have demonstrated antinociceptive (pain-reducing) properties in models of inflammatory, neuropathic, and acute pain. Many of these effects appear to involve suppression of pro-inflammatory cytokines. In neuropathic pain models, ghrelin administration in the spinal cord dampened signaling pathways that drive pro-inflammatory cytokine upregulation. In acute pain models, blocking opioid receptors diminished ghrelin’s analgesic effects, suggesting crosstalk between ghrelin and central opioid systems.
More recent work used a visceral hypersensitivity model that did not involve active inflammation. Using a low concentration of acetic acid to induce transient visceral hypersensitivity, investigators showed that ghrelin mimetics reduced both visceral and somatic hypersensitivity. Blocking ghrelin receptors abolished this effect, confirming it is receptor-mediated. Together, these findings indicate that ghrelin mimetics such as Ipamorelin can relieve pain and hypersensitivity even in the absence of overt inflammation, likely through a combination of anti-inflammatory and central neuromodulatory mechanisms. These results support ongoing interest in ghrelin agonists for GI and abdominal pain states.
Tesamorelin
Tesamorelin has not been directly studied for analgesic effects, and as a GHRH analog, its activity at ghrelin receptors is not expected. However, Tesamorelin does appear to modulate inflammatory pathways. In some studies, Tesamorelin reduced circulating levels of 13 proteins tied to cytotoxic T-cell and monocyte activation, including chemokines such as CCL3, CCL4, CCL13 (MCP-4), and IL-8, as well as cytokines IL-10 and CSF-1 and several T-cell–associated markers (CD8A, CRTAM, GZMA, ADGRG1), plus ARG1, Gal-9, and HGF.[8] Many of these mediators are implicated in neuropathic pain and autoimmune or degenerative conditions with a pain component. Thus, Tesamorelin’s immunomodulatory profile could have indirect relevance for pain, although this remains to be tested directly.
Tesamorelin vs Ipamorelin: Research Notes
At first glance, Tesamorelin might appear to be the more convenient peptide for research because of its FDA approval. Ipamorelin, however, offers important advantages. It is roughly two orders of magnitude less expensive than Tesamorelin, making it very attractive for budget-constrained studies. Ipamorelin has advanced through at least phase II human trials and has a long history in animal research, so safety and dosing are relatively well characterized. In practice, obtaining approval to use either peptide in research should be manageable.
Tesamorelin vs Ipamorelin: Summary
Tesamorelin and Ipamorelin share many similarities but also exhibit meaningful differences. In head-to-head comparison, Tesamorelin appears to have an edge for visceral fat loss and shows promise for cardiovascular and metabolic improvements, though more evidence is needed. Ipamorelin stands out for its impact on bone quality, and emerging data suggest benefits for sleep and cognition. Both peptides are under investigation for a range of potential therapeutic roles.
Two of the clearest distinctions lie in muscle density and pain modulation. Tesamorelin is particularly effective at increasing muscle density by expanding muscle fiber area, making it attractive for addressing age-related muscle loss and functional decline.[6],[7] Ipamorelin is a potent modulator of both inflammatory and non-inflammatory pain through ghrelin receptors, and it is being explored as a tool for GI and visceral pain syndromes.
Ongoing research will clarify the full benefits and limitations of both Tesamorelin and Ipamorelin. What is already apparent is that their actions extend far beyond simple fat reduction. These peptides are helping to refine our understanding of fat quality, metabolic health, and the complex interplay between GH, adipose tissue, muscle, bone, and the nervous system. By doing so, they are opening new avenues for investigating metabolism, energy balance, and aging.
Resources
[1] Clinical Review Report: Tesamorelin (Egrifta). in CADTH Common Drug Reviews. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health, 2016. Accessed: Apr. 06, 2022. [Online]. Available: http://www.ncbi.nlm.nih.gov/books/NBK539131/
[2] P. B. Johansen, Y. Segev, D. Landau, M. Phillip, and A. Flyvbjerg, “Growth hormone (GH) hypersecretion and GH receptor resistance in streptozotocin diabetic mice in response to a GH secretagogue,” Exp. Diabesity Res., vol. 4, no. 2, pp. 73–81, Jun. 2003, doi: 10.1155/EDR.2003.73.
[3] J. V. Gobburu, H. Agersø, W. J. Jusko, and L. Ynddal, “Pharmacokinetic-pharmacodynamic modeling of ipamorelin, a growth hormone releasing peptide, in human volunteers,” Pharm. Res., vol. 16, no. 9, Art. no. 9, Sep. 1999, doi: 10.1023/a:1018955126402.
[4] F. G. De Carvalho, J. N. Justice, E. C. de Freitas, E. E. Kershaw, and L. M. Sparks, “Adipose Tissue Quality in Aging: How Structural and Functional Aspects of Adipose Tissue Impact Skeletal Muscle Quality,” Nutrients, vol. 11, no. 11, p. 2553, Oct. 2019, doi: 10.3390/nu11112553.
[5] J. E. Lake et al., “Tesamorelin improves fat quality independent of changes in fat quantity,” AIDS Lond. Engl., vol. 35, no. 9, pp. 1395–1402, Jul. 2021, doi: 10.1097/QAD.0000000000002897.
[6] S. Adrian et al., “The Growth Hormone Releasing Hormone Analogue, Tesamorelin, Decreases Muscle Fat and Increases Muscle Area in Adults with HIV,” J. Frailty Aging, vol. 8, no. 3, pp. 154–159, 2019, doi: 10.14283/jfa.2018.45.
[7] O. Addison, R. L. Marcus, P. C. Lastayo, and A. S. Ryan, “Intermuscular fat: a review of the consequences and causes,” Int. J. Endocrinol., vol. 2014, p. 309570, 2014, doi: 10.1155/2014/309570.
[8] F. A. White, H. Jung, and R. J. Miller, “Chemokines and the pathophysiology of neuropathic pain,” Proc. Natl. Acad. Sci., vol. 104, no. 51, pp. 20151–20158, Dec. 2007, doi: 10.1073/pnas.0709250104.

Thymosin Alpha 1 10MG
Thymosin Alpha-1 (TA-1) 10mg is a potent, high-dose, lyophilized peptide designed to modulate and enhance immune function. Primarily used to boost T-cell activity, reduce inflammation, and improve responses to infections or chronic illnesses, it is commonly administered via subcutaneous injections. It offers therapeutic support for antiviral defense, autoimmunity, and cancer recovery.
Key Benefits of Thymosin Alpha 1
Immune System Modulation: Enhances T-cell function and boosts natural killer (NK) cells to improve pathogen elimination.
Antiviral & Antibacterial: Utilized in treating chronic infections like Hepatitis B/C, Epstein-Barr, and supporting recovery from sepsis and COVID-19.
Reduced Inflammation & Oxidative Stress: Helps lower markers of chronic inflammation and protects against cellular damage.
Cancer Support: Used as an adjuvant to chemotherapy or radiation to strengthen immune resilience.
Improved Energy: Potential for improved energy levels and vaccine responsiveness.
Side Effects and Safety
Thymosin Alpha-1 is generally well-tolerated. Potential side effects are rare and usually mild, including:
Redness, pain, or irritation at the injection site.
Rare instances of discomfort, such as muscle atrophy or joint pain.
A transient increase in liver enzyme levels (ALT).
Administration and Dosage (10mg formulation)
Format: Supplied as a powder, typically requiring reconstitution with bacteriostatic water before injection.
Dosage: Common, medically supervised protocols involve subcutaneous injection, often in the abdomen or thigh. Dosing varies widely (e.g., 1.5mg, 2-3 times per week) based on the condition being treated.
Storage: Should be stored in a cool, dry place, often refrigerated after reconstitution.
Disclaimer: Thymosin Alpha-1 is a powerful immunomodulator. It is essential to consult a healthcare provider for appropriate dosing and to ensure it is suitable for your health condition.

TIRZ 30
CAS Number: 2023788-19-2
T30 IS a research peptide that helps scientists study how the body manages energy and metabolism. It activates GLP-1 and GIP receptors to show how these signals influence insulin, fat use, and energy levels.
All products are shipped in lyphollized or powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.
Tirzepatide – Research Overview
Class: Dual incretin receptor agonist
Targets:
GIP receptor (GIPR) – glucose-dependent insulinotropic polypeptide
GLP-1 receptor (GLP-1R) – glucagon-like peptide-1
Structure: Synthetic peptide with fatty-acid side chain for extended half-life
Research focus: Metabolic regulation, incretin signaling, insulin pathways, glucagon suppression
Tirzepatide is a dual incretin mimetic designed to activate both GIP and GLP-1 receptors.
It has been studied in rodent and non-human primate research models for its effects on:
- Glucose control
- Energy balance
- Appetite-regulation pathways
- Body-weight regulation
- Incretin hormone signaling
- Insulin secretion and glucagon suppression
All findings refer strictly to preclinical data, not established human outcomes.
Mechanism of Action (Preclinical)
Tirzepatide works by stimulating two incretin receptors simultaneously, creating a multi-layered metabolic signal.
- GIPR Activation (Primary Pathway)
GIP plays major roles in:- Insulin secretion (increased insulin when glucose is high)
- Lipid metabolism
- Adipocyte signaling (fat-cell hormone communication)
Preclinical effects:
- Enhanced glucose-dependent insulin release
- Improved insulin sensitivity
- Reduced adipose inflammation (less inflammatory signaling in fat tissue)
- GLP-1R Activation (Secondary Pathway)
- GLP-1 regulates:
- Insulin secretion
- Glucagon suppression
- Gastric emptying
- Appetite signaling
Preclinical effects:
- Reduced glucagon output
- Slower gastric emptying (food leaves stomach more slowly)
- Lower appetite-signaling activity in the hypothalamus
- Improved glucose tolerance
- Dual-Incretin Synergy
- The key feature of tirzepatide is combined activation of GIPR + GLP-1R.
- In preclinical metabolic studies, dual action has shown:
- Greater metabolic effect than either pathway alone
- More robust insulinotropic response
- Improved energy expenditure markers
- Synergistic appetite-regulation signals
(The “twin incretin” activity is stronger than GLP-1 alone in most rodent models.)
Metabolic Effects Observed in Preclinical Models
-
Glucose Homeostasis
Rodent studies show:- Increased glucose-dependent insulin secretion
- Reduced fasting and post-meal glucose excursions
- Improved β-cell stress markers (better pancreatic function)
- Decreased glucagon levels during hyperglycemia
- Enhanced whole-body insulin sensitivity
-
Body-Weight Regulation Pathways
- Preclinical findings include:
- Reduced food intake
- Lower activation of hypothalamic hunger pathways (ghrelin + AGRP neuron reduction)
- Increased energy expenditure in brown adipose tissue
- Higher fatty-acid oxidation (fat burning)
- Reduced lipogenesis (new fat stored)
-
Adipose Tissue Biology
In rodent adipocyte studies:- Lower inflammation in fat depots (reduced IL-6, TNF-α)
- Improved adiponectin signaling (fat-cell hormone that improves metabolism)
- Reduced hypertrophic adipocytes (smaller fat cells)
-
Liver & Lipid Findings
- Tirzepatide has been evaluated in models of:
- Hepatic fat accumulation
- Lipotoxicity
- Nonalcoholic fatty liver markers
Preclinical results show:
- Reduced triglyceride accumulation
- Improved hepatic insulin signaling
- Better mitochondrial β-oxidation of fats
- Efficacy Observed in Preclinical Studies
- Metabolic Profile
- Strong insulinotropic response when glucose is elevated
- Greater suppression of glucagon compared to single-pathway incretins
- Improved glucose disposal rates
- Lower inflammatory cytokine markers
Appetite & Weight
- Reduced caloric intake
- Lower preference for high-fat diets in rodent choice studies
- Increased satiety signaling
- Significant weight-loss effects in diet-induced obese animals
Cardiometabolic Markers
- Lowered triglycerides and cholesterol in rodent models
- Improved endothelial NO signaling (supports vascular tone)
Reduced oxidative stress markers
(All results from animal/cell studies, not human therapy.)
Safety & Tolerability (Preclinical)
- Generally well-tolerated in animal models
- GI slowing observed due to GLP-1R activity
- No major organ toxicity at research doses
- Long-term safety and PK/PD in humans remain unestablished
- Not FDA-approved for research outside regulated clinical programs
Regulatory Status & Disclaimer
- Tirzepatide is an investigational research peptide.
- It is not approved by the FDA or any regulatory agency for therapeutic, dietary, or cosmetic use.
- For Research Use Only.
- Not for human consumption.
- Not for veterinary use.
- Not for diagnostic or therapeutic applications.
- No medical claims are made or implied.
- Researchers are responsible for appropriate use under local regulation.
Key References (Preclinical)
Wilson J. Dual GIP/GLP-1 receptor activation in metabolic regulation. Cell Metabolism.
Coskun T. Mechanistic profile of tirzepatide in rodent models. Diabetes.
Samms R. GIP receptor biology and energy balance. Endocrinology.
Pfluger P. Incretin hormone synergy and metabolic control. Nat Rev Endocrinol.
Gribble F. Gut peptide signaling in metabolism. J Physiol.
Storage: Peptides should be stored in a dry, cool, dark place. For best preservation, store at 4°C or colder away from bright light.

Tirz 60MG
CAS Number: 2023788-19-2.
Description
T60 is a specialized compound for advanced in vitro metabolic research. Its scientific value is derived from its role as a dual agonist, possessing a strong affinity for both the GLP-1 and GIP receptors. This allows for the comprehensive study of the synergistic relationship between these two critical hormone pathways. Researchers can use T60 to examine how the combined activation of these receptors influences cellular responses, specifically in relation to insulin production, fat utilization, and energy expenditure. The compound offers a stable and consistent model for mapping the intricate hormonal feedback loops that are central to understanding energy management and metabolic health.
All products are shipped in lyphollized or powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.
Tirzepatide – Research Overview
Class: Dual incretin receptor agonist
Targets:
GIP receptor (GIPR) – glucose-dependent insulinotropic polypeptide
GLP-1 receptor (GLP-1R) – glucagon-like peptide-1
Structure: Synthetic peptide with fatty-acid side chain for extended half-life
Research focus: Metabolic regulation, incretin signaling, insulin pathways, glucagon suppression
Tirzepatide is a dual incretin mimetic designed to activate both GIP and GLP-1 receptors.
It has been studied in rodent and non-human primate research models for its effects on:
- Glucose control
- Energy balance
- Appetite-regulation pathways
- Body-weight regulation
- Incretin hormone signaling
- Insulin secretion and glucagon suppression
All findings refer strictly to preclinical data, not established human outcomes.
Mechanism of Action (Preclinical)
Tirzepatide works by stimulating two incretin receptors simultaneously, creating a multi-layered metabolic signal.
- GIPR Activation (Primary Pathway)
GIP plays major roles in:- Insulin secretion (increased insulin when glucose is high)
- Lipid metabolism
- Adipocyte signaling (fat-cell hormone communication)
Preclinical effects:
- Enhanced glucose-dependent insulin release
- Improved insulin sensitivity
- Reduced adipose inflammation (less inflammatory signaling in fat tissue)
- GLP-1R Activation (Secondary Pathway)
- GLP-1 regulates:
- Insulin secretion
- Glucagon suppression
- Gastric emptying
- Appetite signaling
Preclinical effects:
- Reduced glucagon output
- Slower gastric emptying (food leaves stomach more slowly)
- Lower appetite-signaling activity in the hypothalamus
- Improved glucose tolerance
- Dual-Incretin Synergy
- The key feature of tirzepatide is combined activation of GIPR + GLP-1R.
- In preclinical metabolic studies, dual action has shown:
- Greater metabolic effect than either pathway alone
- More robust insulinotropic response
- Improved energy expenditure markers
- Synergistic appetite-regulation signals
(The “twin incretin” activity is stronger than GLP-1 alone in most rodent models.)
Metabolic Effects Observed in Preclinical Models
-
Glucose Homeostasis
Rodent studies show:- Increased glucose-dependent insulin secretion
- Reduced fasting and post-meal glucose excursions
- Improved β-cell stress markers (better pancreatic function)
- Decreased glucagon levels during hyperglycemia
- Enhanced whole-body insulin sensitivity
-
Body-Weight Regulation Pathways
- Preclinical findings include:
- Reduced food intake
- Lower activation of hypothalamic hunger pathways (ghrelin + AGRP neuron reduction)
- Increased energy expenditure in brown adipose tissue
- Higher fatty-acid oxidation (fat burning)
- Reduced lipogenesis (new fat stored)
-
Adipose Tissue Biology
In rodent adipocyte studies:- Lower inflammation in fat depots (reduced IL-6, TNF-α)
- Improved adiponectin signaling (fat-cell hormone that improves metabolism)
- Reduced hypertrophic adipocytes (smaller fat cells)
-
Liver & Lipid Findings
- Tirzepatide has been evaluated in models of:
- Hepatic fat accumulation
- Lipotoxicity
- Nonalcoholic fatty liver markers
Preclinical results show:
- Reduced triglyceride accumulation
- Improved hepatic insulin signaling
- Better mitochondrial β-oxidation of fats
- Efficacy Observed in Preclinical Studies
- Metabolic Profile
- Strong insulinotropic response when glucose is elevated
- Greater suppression of glucagon compared to single-pathway incretins
- Improved glucose disposal rates
- Lower inflammatory cytokine markers
Appetite & Weight
- Reduced caloric intake
- Lower preference for high-fat diets in rodent choice studies
- Increased satiety signaling
- Significant weight-loss effects in diet-induced obese animals
Cardiometabolic Markers
- Lowered triglycerides and cholesterol in rodent models
- Improved endothelial NO signaling (supports vascular tone)
Reduced oxidative stress markers
(All results from animal/cell studies, not human therapy.)
Safety & Tolerability (Preclinical)
- Generally well-tolerated in animal models
- GI slowing observed due to GLP-1R activity
- No major organ toxicity at research doses
- Long-term safety and PK/PD in humans remain unestablished
- Not FDA-approved for research outside regulated clinical programs
Regulatory Status & Disclaimer
- Tirzepatide is an investigational research peptide.
- It is not approved by the FDA or any regulatory agency for therapeutic, dietary, or cosmetic use.
- For Research Use Only.
- Not for human consumption.
- Not for veterinary use.
- Not for diagnostic or therapeutic applications.
- No medical claims are made or implied.
- Researchers are responsible for appropriate use under local regulation.
Key References (Preclinical)
Wilson J. Dual GIP/GLP-1 receptor activation in metabolic regulation. Cell Metabolism.
Coskun T. Mechanistic profile of tirzepatide in rodent models. Diabetes.
Samms R. GIP receptor biology and energy balance. Endocrinology.
Pfluger P. Incretin hormone synergy and metabolic control. Nat Rev Endocrinol.
Gribble F. Gut peptide signaling in metabolism. J Physiol.
Quality & Specifications
We understand that reliable and reproducible data depends on the quality of your reagents.
Manufacturing Standards: Our T60 is synthesized under strict quality control protocols to ensure high purity and batch-to-batch consistency.
Independent Verification: Every lot is validated by an independent, third-party laboratory to confirm its identity and purity.
Certificate of Analysis: A comprehensive Certificate of Analysis (CoA) is included with every shipment, detailing the specific test results for your batch.
Optimized Stability: The peptide is supplied in a lyophilized (freeze-dried) powder form. This process removes water without damaging the peptide structure, ensuring maximum stability and extending its shelf life.
Preparation: Prior to use, the powder must be reconstituted with a research-grade sterile solvent (not included) to ensure accurate and repeatable concentrations for your experiments.
Research Use Only
This product is intended strictly for laboratory and in vitro research applications. T60 is NOT for human or veterinary use, diagnostic purposes, or therapeutic applications. Must be handled by qualified professionals in a controlled setting.
Product Care:
Store in a cool, dry place away from light. If reconstituted, Please Refrigerate. For longer term storage, freezing at -20°C is recommended to maintain integrity.
Product Note:
All products are shipped in lyophilized or original powder form and must be reconstituted to a liquid for research and testing. We are unable to provide any dosing instructions, however all products should be considered pharmaceutical grade.


VIP 5MG (Vasoactive Intestinal Peptide)
Research:anti-inflammatory, immunity
A VIP bioregulator is vasoactive intestinal polypeptide (VIP), a neuropeptide that acts as both a neurotransmitter and a hormone, playing a key role in regulating bodily functions. It has widespread effects, including relaxing smooth muscles, modulating the immune system, regulating hormone secretion, and influencing water and electrolyte balance in the gastrointestinal tract. VIP (vasoactive intestinal peptide) is a 28-amino acid hormone peptide that regulates smooth muscle function and cellular secretion across multiple biological systems.
Functions of VIP (used to research the following)
- Vascular and smooth muscle: VIP is a potent vasodilator, meaning it relaxes blood vessels and other smooth muscles in the respiratory, gastrointestinal, and reproductive tracts.
- Gastrointestinal regulation: It regulates water and electrolyte secretion from the pancreas and gut, and influences the release of pancreatic hormones.
- Immune system modulation: VIP helps modulate the immune system and can decrease the expression of inflammatory factors.
- Neuroendocrine function: It acts as a neurohormone and a neurotransmitter, affecting the central and peripheral nervous systems and influencing the release of hormones like prolactin from the pituitary gland.
- Other functions: It also promotes vaginal lubrication, stimulates exocrine and endocrine secretion, and can affect cell proliferation
Vasoactive Intestinal Peptide (VIP) – Research Overview
Full name: Vasoactive Intestinal Peptide
Class: Neuropeptide / Regulatory peptide
Sequence: 28-amino acid peptide
Primary targets: VPAC1 and VPAC2 receptors
Research focus: Neuroimmune signaling, smooth-muscle relaxation, vasodilation, circadian pathways, anti-inflammatory signaling
VIP is an endogenous peptide widely distributed in the brain, lungs, gut, and immune system.
It has been extensively studied in preclinical models for its effects on:
-Immune modulation
-Anti-inflammatory pathways
-Smooth muscle relaxation
-Pulmonary biology
-Neurological signaling
-Vascular tone regulation
All findings refer to animal and cellular research, not established human outcomes.
Molecular & Pharmacological Profile
VIP belongs to the secretin/glucagon peptide family.
In preclinical systems, it binds to two major G-protein–coupled receptors:
VPAC1
(expressed in immune cells, lungs, gut tissue)
VPAC2
(expressed in CNS, endocrine tissue, and smooth muscle)
Activation of these receptors is associated with:
- Potent vasodilation (blood-vessel relaxation)
- Bronchodilation (airway relaxation)
- Inhibition of inflammatory cytokines
- Upregulation of cyclic AMP (cAMP) (increases intracellular signaling)
- Modulation of circadian rhythm via suprachiasmatic nucleus pathways
- Neuroprotection markers in neuronal models
Mechanisms of Action (Preclinical)
-
cAMP Elevation
VIP strongly increases intracellular cAMP
(a messenger that drives anti-inflammatory + relaxation pathways).
This leads to:- Smooth muscle relaxation
- Suppression of inflammation
- Neuroprotective responses
-
Immune Modulation
VIP reduces:
TNF-α
-(IL-6, IL-12)
Interferon-γ
(markers of inflammation in preclinical immune studies)
And increases:
IL-10 (anti-inflammatory cytokine) -
Smooth Muscle Relaxation
VIP causes rapid relaxation of:- Vascular smooth muscle
- Airway smooth muscle
- Gastrointestinal smooth muscle
(linked to its vasodilatory + bronchodilatory effects in animals)
-
Neuroendocrine & Circadian Effects
VIP is a key neuropeptide in the suprachiasmatic nucleus (SCN)
(the brain’s master circadian clock)
where it regulates:- Sleep/wake cycle gene expression
- Neural synchrony
- Clock-protein rhythmicity
-
Neuroprotective Pathways
Preclinical data show:- Reduced oxidative stress in neuronal cultures
- Increased neuronal survival after glutamate challenge
- Anti-apoptotic signaling (cell-survival pathways)
- Preclinical Research: Potential Applications
VIP has been evaluated in animal and cell-based models related to:
-
Pulmonary & Airway Research
Smooth-muscle relaxation (bronchodilation)
Reduced airway inflammation
Modulation of immune cells in the lung -
Vascular & Cardiovascular Models
Vasodilation (blood-vessel relaxation)
Increased blood flow in preclinical systems
Reduced vascular resistance -
Neuroimmune & Inflammatory Pathways
Lower inflammatory cytokine production
Regulation of T-cell activity
Inhibition of macrophage activation
(supportive of immune balance in experimental models) -
Neurological & Cognitive Studies
Neuroprotective responses under stress
Influence on SCN circadian signaling
Support of neuronal survival
(preclinical findings — not human therapeutic claims) -
Gastrointestinal Models
Smooth muscle relaxation
Regulation of intestinal motility
Support of mucosal integrity in rodent models
Efficacy Observed in Preclinical Studies
Anti-Inflammatory Effects
Reduced TNF-α, IL-6, and IL-12
Increased IL-10
(strong anti-inflammatory pattern)
Vascular + Pulmonary Findings
Significant relaxation of airway smooth muscle
Increased bronchodilation in guinea-pig models
Enhanced regional blood flow in vascular research
Neuroprotective & Circadian Outcomes
Improved neuronal viability
Modulation of clock genes
Enhanced circadian rhythm stability
Immune System Regulation
Suppressed macrophage and dendritic-cell activation
Balanced Th1/Th2 signaling in rodent studies
These effects appear dose-dependent across multiple models.
Safety & Tolerability (Preclinical)
VIP generally shows low toxicity in rodent studies
Rapid degradation in plasma (short half-life)
Minimal off-target hormonal activation
Most effects linked to cAMP-mediated signaling
Long-term safety in humans has not been established.
VIP is not FDA-approved for therapeutic use.
Regulatory Status & Disclaimer
- Vasoactive Intestinal Peptide (VIP) is an investigational research compound.
- It is not approved by the FDA or any regulatory agency for therapeutic, dietary, or cosmetic use.
- For Research Use Only.
- Not for human consumption.
- Not for veterinary use.
- Not for diagnostic or therapeutic purposes.
- No medical claims are made or implied.
- Researchers must follow all applicable regulations.
Key References (Preclinical)
Gozes I. VIP as a neuroprotective peptide. Front Endocrinol.
Delgado M. VIP and immune modulation in preclinical models. J Immunol.
Said S. Vasoactive intestinal peptide and smooth muscle relaxation. Am J Physiol.
Brenneman D. VIP-mediated neuronal survival pathways. Peptides.
Harmar A. VIP and circadian rhythm regulation. Proc Natl Acad Sci.
Our pharmaceutical-grade VIP maintains high purity standards for reliable research outcomes. Each batch undergoes rigorous testing to support accurate scientific investigations of this important regulatory peptide’s mechanisms and potential therapeutic applications in laboratory research use settings.
Each vial contains 5 mg of lyophilized VIP. Reconstitute immediately before research use in bacteriostatic water, aliquot single-use, and store at ≤ –20 °C to avoid repeated freeze–thaw cycles.